Immune support is set to be a clear winner from the recently published article 13.1 general health claims in the EU. 10 ingredients have won immunity claims: Copper, folate, iron, selenium, vitamins A, B12, B6, C, D and zinc.
Global retail value of immune support in food-beverage products and excluding food supplements stood at US$2.6 billion in 2012, 61% of which was driven by the EU, and globally is set to see an additional US$80 million in growth over 2012-2017. (The figure is for products making explicit immunity claims).
With a clear set of authorised ingredients that can be used in a wide variety of products these claims can significantly help towards reversing the decline of US$29 million forecast for the EU over 2012-2017.
The big 10 – a path to “relatively cheap innovation”
Article 13.1 general health claims, published in May 2012 under the EU nutrition and health claims regulation (NHCR), opens opportunities for the relatively cheap innovation and repositioning of products, using simple ingredients such as vitamins or minerals, to target immune support. From the 222 approved article 13.1 general health claims, immune support gained 11 claims for the 10 nutrients above.
A number of brands have been quick to make use of this list of ingredients. In the UK GlaxoSmithKline has widened its product line with Ribena Plus containing vitamins A, C and E targeting immune support for children.

The Boots Company launched a private label drink named Daily Defence, with vitamins B, C and zinc as part of the Shaper’s Watervit range. Elsewhere, Agrosnova’s Dr. Witt ActivDrink from Poland has used the claims to target different health platforms including immunity via the addition of vitamin C into its premium range of drinks, functional gum and nectars.
Immunity claims without probiotics
The key players in immune support have established their products based on the benefits of probiotics.
Yakult’s pioneering probiotic immunity drink was worth US$174mn in the EU in 2011. Danone’s drinking yoghurt, Actimel, sells $1.5bn globally, but that figure is 6% lower than the year before.
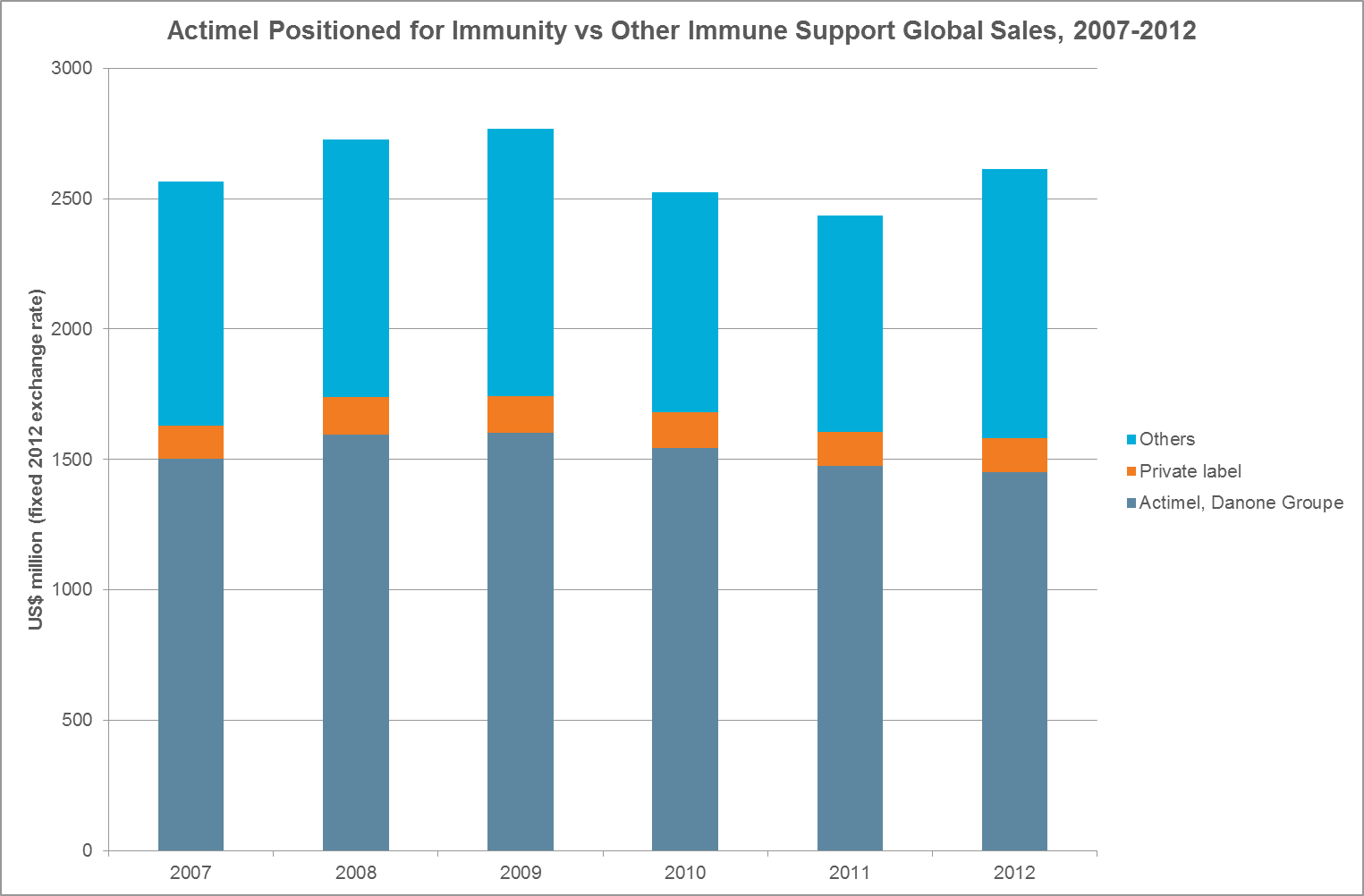
But brands such as Benessere Attivo and Kyr boosted immune support sales with strong growth between 2006-2011.
But no probiotic health claims have been authorised in the EU, and so the temptation to add ingredients from ‘the big 10’ rises. This is exactly the strategy that a number of manufacturers have decided to take.
Actimel has already undergone reformulation in a number of EU countries. Whilst keeping the L casei probiotic culture (as the name of the strain can still be mentioned), it has heavily marketed the fact its product now contains vitamin D and B6, both of which have claims for, “the contribution to the normal function of the immune system”.
But the question arises: If probiotics do not carry a claim is there any benefit to keeping them in a product? In that area, there is manufacturer and consumer faith in their efficacy coupled with the fact there are other channels of health information other than health claims which may explain ongoing global category growth despite the slight EU dip.
Opportunities for immune support in an ageing population
The ageing population has been a driver of growth. The number of individuals aged over 65 in the EU increased by 5m between 2006-2011, providing a large number of new consumers very interested in general wellbeing, as consumers increasingly turn to prevention rather than treatment of ailments.
Therefore, through the use of article 13.1 general health claims manufacturers who see the potential in immune support products either through reformulation or through repositioning their product could see success.
But with so many products expected to introduce these claims in the near future it will be those companies that further invest in research to gain article 13.5 or 14 claims that will really differentiate themselves from their competitors.
This is first of a series of articles that will focus on a number of key health positionings set to be particularly affected by the article 13.1 health claims published in May 2012.
