Probiotic strain may help prevent obesity

The strain has already been reported to curb the absorption of dietary triglyceride (TAG), high blood levels of which are linked to clogged arteries (atherosclerosis) and therefore the risk of heart disease and stroke.
However the results of this latest study suggest Lactobacillus gasseri SBT2055 (LG2055) may also enhance energy expenditure by stimulating carbohydrate oxidation, improve glucose tolerance and reduce levels of the pro-inflammatory mediator serum amyloid P (SAP) in rats.
“Considering that these effects correlate with the reduction of body weight gain, our findings suggest multiple actions activated by LG2055 that may work in synergy to prevent obesity,” the researchers wrote in the British Journal of Nutrition.
The study was conducted by researchers from the Kyushu University, Saga University and Nishikyushu University in Japan along with the Milk Science Research Institute of Megmilk Snow Brand Co., which produces probiotic dairy products.
They induced obesity and glucose intolerance in six rats through diets containing high sucrose and high saturated fat lard and then split the animals between a control group and one given a diet containing LG2055.
The final concentration of viable LG2055 in the fermented skimmed milk-containing diet was 6×107 colony-forming units per gram of diet.
The whats
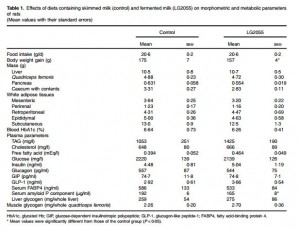
In this study the researchers found the LG2055 diet significantly reduced serum levels of the pro-inflammatory mediator SAP.
The control group’s SAP was 192 micrograms (μg) per ml, while for the probiotic group this sat at 165 μg/ml.
The researchers said this suggested LG2055 could prevent low-grade inflammation associated with obesity and glucose metabolism disorders.
The normal range of SAP in the serum is comparable in rats and humans and ranges from 10–50 μg/ml, meaning the results could be extrapolated to humans, they said.
The whys
The researchers said this anti-obesity effect and improvement in glucose tolerance may be down to improved energy expenditure through an increase in carbohydrate oxidation.
“Mammals utilise not only glucose, long-chain fatty acids and amino acids as energy sources, but also SCFA derived from colonic fermentation of dietary fibre by the gut microbiota,” they wrote.
“The association of microbial activity in the gastrointestinal tract with host energy homoeostasis and obesity pathogenesis is increasingly recognised.”
They said that future studies should investigate whether these effects could be even greater if pre- and probiotics were used in combination.
Source: British Journal of Nutrition
Published online ahead of print, doi:10.1017/S0007114516002245
“Probiotic Lactobacillus gasseri SBT2055 improves glucose tolerance and reduces body weight gain in rats by stimulating energy expenditure”
Authors: B. Shirouchi1 , K. Nagao , M. Umegatani , A. Shiraishi, Y. Morita, S. Kai, T. Yanagita, A. Ogawa, Y. Kadooka and M. Sato