Small intestine bacteria key in fat uptake, suggests mouse data
Published in Cell Host & Microbe, the animal data shows that germ-free mice lacking in gut bacteria, were unable to digest or absorb fatty foods when fed a high-fat diet.
The microbiome-free mice also failed to put on weight and produced fatty stools, said the University of Chicago team.
In contrast, specific pathogen free (SPF) mice - hich had a healthy gut microbiota but lack certain disease causing species - were found to quickly gain weight in response to a high fat diet.
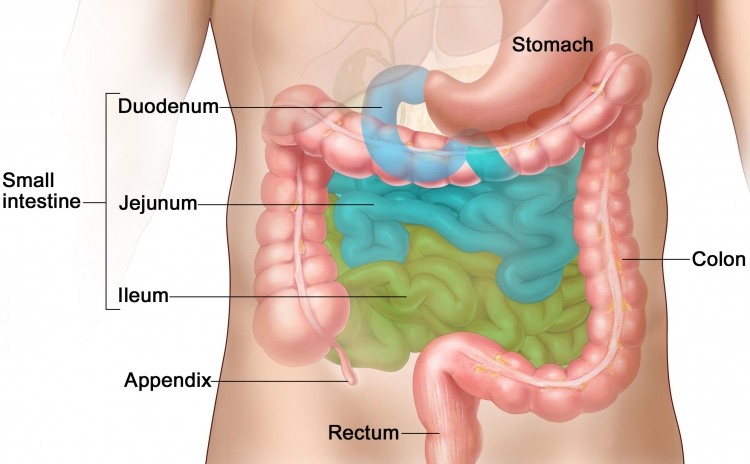
“Few people have focused on the microbiome of the small intestine, but this is where most vitamins and other micronutrients are digested and absorbed," said senior author Professor Eugene Chang from the University of Chicago.
"Our study is one of the first to show that specific small-bowel microbes directly regulate both digestion and absorption of lipids," he added. "This could have significant clinical applications, especially for the prevention and treatment of obesity and cardiovascular disease."
According to Chang and colleagues, the high-fat diet (HFD) also rapidly increased the abundance of certain types of bacteria (Clostridiaceae and Peptostreptococcaceae), while decreasing the numbers of others (Bifidobacteriacaea and Bacteriodacaea).
Reduced levels of the latter two bacteria have been associated with obesity, the team noted.
Bacterial transfer
In addition to finding certain bacterial species that were linked to the high-fat diet, the team also looked at the effect of microbial transfer the SPF mice to the germ-free mice.
Indeed, when the team introduced bacteria from the jejunum part of the small intestine of SPF mice into germ-free mice, the previously fat-immune recipients rapidly gained the ability to absorb fats.
Furthermore, germ-free mice who received the bacteria were also found to retain a higher capacity to absorb fat even when placed onto a low fat diet, said the authors.
"Our study found that, at least in mice, a high-fat diet can profoundly alter the microbial make-up of the small intestine,"
"Certain dietary pressures, such as calorie-dense foods, attract specific bacterial strains into the small intestine," the lead author said.
"These microbes are then able to allow the host to digest this high-fat diet and absorb fats. That can even impact extra-intestinal organs such as the pancreas."
The team said efforts to manipulating the microbiota and alter the relative abundance of bacteria that either promote or inhibit fat absorption in the small intestine could form part of future strategies to battle obesity.
Furthermore, the technique could also have benefits for people with conditions that involve malabsorption is present.
"Maybe we could use pre- or probiotics or even develop post-biotics (bacterial-derived compounds or metabolites) to enhance nutrient uptake for people with malabsorption disorders, such as Crohn's disease, or we could test novel ways to decrease obesity," commented first author Assistant Professor Kristina Martinez-Guryn, currently at Midwestern University, Downers Grove, IL.
"I would say the most important takeaway overall is the concept that what we eat--our diet on a daily basis--has a profound impact on the abundance and the type of bacteria we harbour in our gut," added Martinez-Guryn. "These microbes directly influence our metabolism and our propensity to gain weight on certain diets."
Source: Cell Host & Microbe
Volume 23, pages 458-469, published online 11 April 2018, doi:10.1016/j.chom.2018.03.011
"Small intestine microbiota regulate host digestive and absorptive adaptive responses to dietary lipids,"
Authors: Kristina Martinez-Guryn, Eugene B. Chang et al