International study links specific gut microbes with common illnesses and obesity
NutraIngredients reported the preliminary results of the large-scale PREDICT 1 study last summer. Now, the full results of this, plus the PREDICT 2 primary investigation, are published in the journal 'Nature Medicine'.
The study team - including researchers from ZOE, King’s College London, the Harvard T.H. Chan School of Public Health, Massachusetts General Hospital (MGH), and the University of Trento, Italy - say these findings can be used to provide personalised dietary advice for better health, based on gut microbiome testing.
They analysed detailed data on the composition of participants’ gut microbiomes, their dietary habits, and cardiometabolic blood biomarkers. They gathered microbiome sequence data, detailed long-term dietary information, and results of hundreds of cardiometabolic blood markers from just over 1,100 participants in the U.K. The PREDICT 2 then completed its primary investigations in 2020 with a further 1,000 U.S participants, and PREDICT 3 launched a few months ago.
The results show that the microbes found in the gut are directly linked with specific foods and that the microbiome has a greater association to biomarkers of metabolic disease than other factors, such as genetics.
'Good' and 'bad' bugs
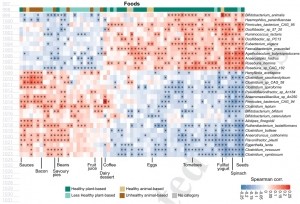
They found clear segregation of microbes into two distinct clusters associated with either more healthy plant-based foods (such as spinach, seeds, tomatoes, broccoli) or less healthy plant-based (juices, sweetened beverages, refined grains) and animal-based foods.
Taxa linked to healthy plant-based foods mostly included butyrate producers, such as Roseburia hominis, Agathobaculum butyriciproducens, Faecalibacterium prausnitzii and Anaerostipes hadrus, as well as uncultivated species, predicted to have this metabolic capability (Roseburia bacterium CAG:182 and Firmicutes bacterium CAG:95).
Clades correlating with less healthy plant-based and animal-based foods included several Clostridium species (Clostridium innocuum, Clostridium symbiosum, Clostridium spiroforme, Clostridium leptum, Clostridium saccharolyticum).
The segregation of species according to animal-based healthy foods (eggs, white and oily fish) or animal-based less healthy foods (meat pies, bacon, dairy desserts) was also distinct and overlapping with taxa signatures for healthy and less healthy plant foods.
The diversity and quality of a healthy diet was particularly predictable by the microbiome, highlighting the importance of looking beyond nutrients and single foods in diet–microbiome research.
Dr. Sarah Berry, Reader in Nutrition Sciences at King’s College London and co-author of the paper said, “As a nutritional scientist, finding novel microbes that are linked to specific foods, as well as metabolic health, is exciting. Given the highly personalised composition of each individuals’ microbiome, our research suggests that we may be able to modify our gut microbiome to optimise our health by choosing the best foods for our unique biology.”
Nicola Segata, PhD, professor and principal investigator of the Computational Metagenomics Lab at the University of Trento, Italy and leader of the microbiome analysis in the study, said:“We were surprised to see such large, clear groups of what we informally call ‘good’ and ‘bad’ microbes emerging from our analysis.
“It is also exciting to see that microbiologists know so little about many of these microbes that they are not even named yet. This is now a big area of focus for us, as we believe they may open new insights in the future into how we could use the gut microbiome as a modifiable target to improve human metabolism and health.”
The report notes that future microbiome studies should undertake more detailed dietary assessment methodologies, rather than always concentrating on specific nutrients.
"The substantially greater detail and consistency in our results relative to previous diet–microbiome work may be due to the quality in dietary recording, metagenomic profiling and the large sample size. However, given the limitations of FFQ dietary data, future diet–microbiome studies would benefit further from higher resolution dietary assessment methodologies, such as weighed food record data."
Link with obesity
The researchers also found that the gut microbiome is a better predictor of postprandial triglycerides and insulin concentrations than of glucose levels.
They report reveals robust microbiome-based biomarkers of obesity, as well as markers for cardiovascular disease and impaired glucose tolerance, which are key risk factors for COVID.
It states: "Our machine learning approach found visceral fat to be more strongly linked to gut microbial composition than BMI33, a finding again validated in US participants. Some obesity-associated taxa were also indicators of poor dietary patterns after controlling for BMI (for example, Clostridium CAG:58, Flavonifractor plautii), whereas markers of lower visceral fat mass (for example, F. prausnitzii) were more strongly linked to healthier foods and patterns of intake, illustrating that diet and obesity microbiome signatures overlap but are not identical."
Personalised nutrition
Health science start-up company ZOE has already translated these cutting-edge discoveries into an at-home microbiome test, currently available in the US and to be made available in the UK by the end of the year.
The study's coordinator Professor Tim Spector, Epidemiologist from King’s College London and scientific founder of ZOE explains, says: “I am very excited that we have been able to translate this cutting edge science into an at-home test in the time it has taken for the research to be peer reviewed and published.
“Through ZOE, we can now offer everyone an opportunity to discover which of these microbes they have living in their gut. By using machine learning, we are able to share with you our calculations of how your body will respond to any food.”
Source: Nature Medicine
"Microbiome connections with host metabolism and habitual diet 1,098 deeply phenotyped individuals"
https://doi.org/10.1038/s41591-020-01183-8
Segata. N., et al