“It is a sign of our commitment to help grow and change the omega-3 category,” DSM’s global head of marketing communications Outi Helena Armstrong told us.
The Dutch-Swiss firm said oversized omega-3 pills were "a thing of the past."
The process technology it uses to make its new omega-3 line of called MEG-3 Ultra and Life’s DHA Ultra was launched simultaneously at nutraceutical trade show Supply Side West in Las Vegas and pharmaceutical event CPhI in Barcelona last week.
Technology showcase
The technology, which took about two years to develop, uses fractional distillation and vacuum treats the oils in one step, which DSM says ensures the quality of the heat-sensitive oils.
“Instead of multiple separation passes, 3C uses only one. It seperates EPA and DHA much more effectively and it can use fish oils from a wider variety of fish oils around the world,” DSM said of the process which can also be used for algal sources.
With the technology DHA and EPA levels can reach up to 85% compared to up to 70% with DSM’s previous process and DHA:EPA ratio can be tailored, it says. Each Ultra softgel capsule offers 250 mg of EPA (eicosapentaenoic acid) and DHA (docosahexaenoic acid) omega-3.
The waste oil of the process is then used to power its facility in Mulgrave, Nova Scotia.
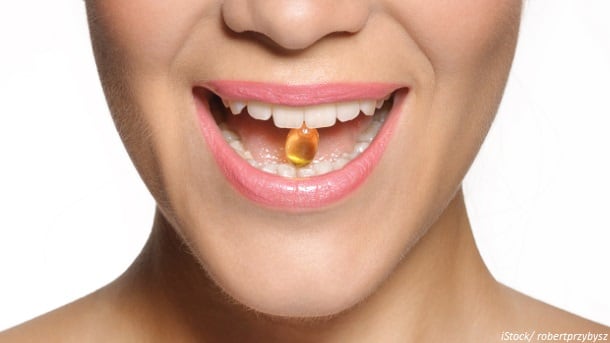
Easy to swallow?
Speaking with us at CPhI, Helena Armstrong said capsule size was a key barrier for use.
“Some of our customers say consumers phone in and say it’s hard to swallow and they’re not going to swallow a big capsule like that. This [3C technology] is obviously a way to address that concern.”
Helena Armstrong added this also held cost-cutting benefits in the amount of material like gelatin needed to make the smaller capsules.
Dr Manfred Eggersdorfer, DSM’s senior vice president of nutrition science and advocacy said this was a particular issue for groups like children and the elderly.
Omega-3 health claims
DHA is perhaps one of the most claim-backed nutrients available in Europe, with approved health claims for brain function, vision, infant development, heart function and blood pressure.
DSM recently applied for a further claim on DHA and memory, but the dossier was rejected by the European Food Safety Authority (EFSA) in May.
Eggersdorfer told us at CPhI the company had been disappointed by the rejection. Yet despite continuing to conduct research in this area, it had no plans to re-apply for now.
"We know that from the other companies in the field there are a number of studies going on which are supportive. So maybe something is coming back."
Helena Armstrong added: "We think that it's quite natural that when science evolves as it does, that it's very evident that health claims evolve in future. They should in principle go hand in hand, but what happens and when is not so clear to predict."
