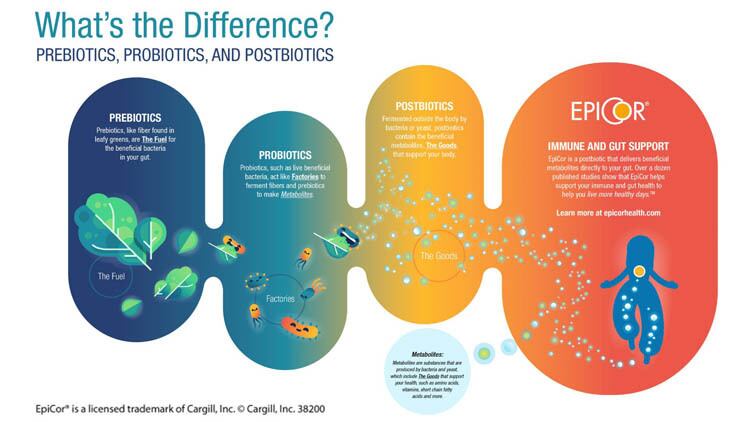
Cargill, well-known for its food and beverage ingredients, is making a strategic splash in the area of human health with the #1 postbiotic brand, EpiCor.
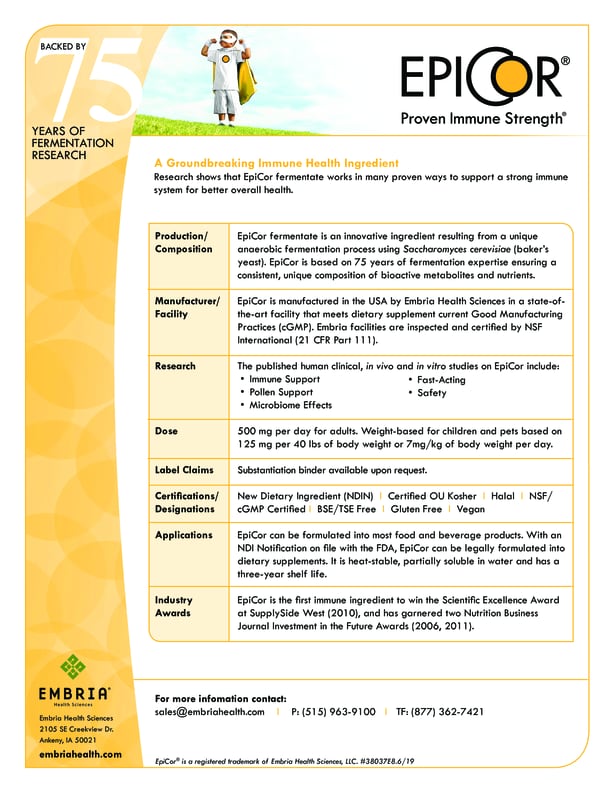
EpiCor postbiotic is an ingredient derived from Saccharomyces cerevisiae, otherwise known as baker’s yeast. The fermentation experts at Cargill use a proprietary process to ensure nothing is filtered out. The result, is a unique fingerprint of metabolites; a rich combination of vitamins, nutrients, amino acids and antimicrobial peptides geared to provide a health benefit.
Baked by over a dozen published studies, EpiCor is clinically shown to support the gut microbiome1, immune health2,3, and nasal comfort4. The research supporting this ingredient includes randomized, double-blind, placebo-controlled human trials with statistically significant outcomes.
EpiCor postbiotic is proud to deliver the beneficial properties consumers want, and the formulation flexibility brands need. Brands can now formulate the science-backed benefits of this pH, heat, and shelf stable ingredient into supplements and functional foods and beverages.
The past few years have been monumental for postbiotics as they’ve gained significant consumer traction and witnessed a surge of clinical research. As consumers look for ways to better support their health and immunity, they may have just found it with postbiotics.
-----
[1] Pinheiro, I., et al. (2017). A yeast fermentate improves gastrointestinal discomfort and constipation by modulation of the gut microbiome: results from a randomized double-blind placebo-controlled pilot trial BMC Complement Altern Med 17, 441.
[2] Moyad MA, et al. Effects of a modified yeast supplement on cold/flu symptoms. Urol Nurs 2008, 28(1):50-55.
[3] Moyad MA, et al. Immunogenic yeast-based fermentate for cold/flu-like symptoms in nonvaccinated individuals. J Altern Complement Med 2010, 16(2):213-218.
[4] Moyad MA, et al. Immunogenic yeast-based fermentation product reduces allergic rhinitis-induces nasal congestion: a randomized, double-blind placebo-controlled trial. Adv Ther 2009, 26(8):795-804.