Teenage microbiome disruption linked to multiple sclerosis trigger: Mouse data
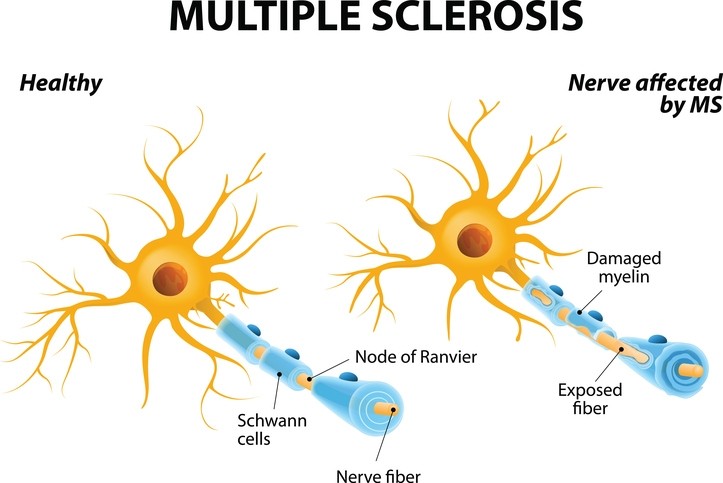
The disruption of gut bacteria composition was found to trigger an MS-like condition in mice known as experimental autoimmune encephalomyelitis (EAE), which is used to model the human disease in animals.
The expansion in numbers of harmful species of gut bacteria cause the breakdown of the immune system’s tolerance to central nervous system (CNS) pathogens, found researchers from the Department of Neurology, Rutgers–Robert Wood Johnson Medical School, Piscataway, New Jersey.
They also discovered that there was a particular age window of increased vulnerability to the disease. During this period, from adolescence to young adulthood, the immature immune system was less able to tolerate attacks from the harmful bacteria.
“We report here that gut dysbiosis, which we attribute to expansion of enteric pathogenic bacteria, triggers and/or exacerbates the spontaneous development of EAE, an animal model of MS,” wrote co-first authors Sudhir Yadav and Sridhar Boppana.
“This study suggests that gut dysbiosis may play a pathological role in the initiation and/or progression of MS during a defined age window,” they added.
"The findings could have therapeutic implications on slowing down MS progression by manipulating gut bacteria," commented Suhayl Dhib-Jalbut, Director of Rutgers-Robert Wood Johnson Centre for Multiple Sclerosis.
Although gut dysbiosis is common in MS patients, the way in which the alteration of the microbiome is involved in disease development has been unknown until now. This study is therefore the first to identify the mechanisms underlying the gut bacteria’s influence on the immune system that may lead to MS.
The results may in future enable the manipulation of the gut microbiota to eliminate pathogenic species involved with MS progression and/or increase numbers of beneficial bacteria as a preventive measure.
Possible mechanism
The mechanism by which pathogenic bacteria may affect the immune system is complex. Three particular genes may be involved, suggest the researchers.
Firstly, the dysbiosis may cause upregulation of the complement C3 gene. This gene regulates the production of a protein which is involved in destroying foreign pathogens such as bacteria. C3 upregulation may in turn lower the activity of the Foxp3 and E3 ubiquitin ligase genes. Lower expression of these genes may lead to the development of autoimmune conditions, suggest the researchers.
In particular, low Foxp3 activity is recognised to reduce T-regulatory cell activity allowing autoimmune cells to attack the body’s own tissues. – a characteristic of MS.
“These data suggest that gut dysbiosis-mediated down-regulation of Foxp3 and E3 ubiquitin ligase genes could be involved in the breakage of immunological tolerance to CNS antigens,” hypothesised the authors.
The findings indicate that a combination of gut bacteria imbalance, age and MS-risk genes may work in collaboration to trigger disease, suggest the researchers.
IPA World Congress + Probiota 2018
The rapidly evolving universe of probiotics, prebiotics and the microbiome will be discussed in-depth at the upcoming IPA World Congress + Probiota 2018 in Barcelona on February 7-9.
From microbiome advances, to start-up game changers, market stats, crucial clinical science and regulatory knowledge, this is a congressional must-have.
Will you be joining your peers in one of Europe’s great cities?
--
Source: Proceedings of the National Academy of Sciences
Volume 114, no. 44, pages E9318–E9327, doi: 10.1073/pnas.1615715114
“Gut dysbiosis breaks immunological tolerance toward the central nervous system during young adulthood”
Authors: Sudhir Yadava, Sridhar Boppana et al.