
EU continues CBD scrutiny, rejecting 6 more novel food applications
The European Commission has terminated a backlog of cannabidiol (CBD) novel food applications for lack of data and non-compliance.

The European Commission has terminated a backlog of cannabidiol (CBD) novel food applications for lack of data and non-compliance.

With making America healthy again at the top of the public agenda, Kyle Diamantas discussed the FDA’s focus on dietary supplement modernization, the reform of ingredient safety pathways and stronger oversight of the global supply chain.

The National Advertising Division (NAD) has recommended that Bayer HealthCare LLC discontinue or modify a series of claims tied to its One A Day Men’s Pre-Conception Health Complete Multivitamin, concluding that product positioning and messaging conveyed...

Formulated supplementary sports foods (FSSF) containing caffeine will need to add new advisory and warning statements, says the Foods Standards Australia New Zealand (FSANZ).

After a disastrous attempt to implement new food safety regulations in January, Vietnam has taken next steps forward — but looks unlikely to make the original April deadline

Global demand for spirulina is booming, with the market for this superfood projected to reach $1.3 billion by 2032. But new testing revealed some cracks in quality and safety that suggest some top-selling products may expose consumers to elevated lead...

Dietary supplement companies may be focused on FDA activity in Washington, but at the Sports & Active Nutrition Summit (SANS), Rend Al‑Mondhiry, partner at Amin Wasserman Gurnani LLP, warned that some of the major risks are emerging much closer to home.


China’s factory registration policy for overseas-made health foods, magtein approval in the UK and EFSA’s public consultation on berberine-containing plants are some of the latest updates in this regulatory news round-up.

Women’s health brands Lunera, Minerva Wellness and Nova breached multiple advertising codes by making claims that their supplements could treat or significantly alleviate menopause symptoms, according to the UK’s Advertising Standards Authority (ASA).

Overseas-made health foods entering China via the general trade route will still require official recommendation letters from their local authority when registering their manufacturing facility with the Chinese authorities - a move an industry expert...

A lawsuit challenging the nutrition claims of David Protein bars is putting calorie counts under the microscope and highlighting a deeper debate over how those numbers are determined in the first place.

The upcoming public Food and Drug Administration (FDA) meeting exploring the scope of dietary supplement ingredients will revisit one of the most contested and, according to DSHEA’s architects, most misunderstood phrases in the U.S. dietary supplement law.

Catch up with our weekly round-up of key news from across the Nutraverse.

Vitamin and mineral supplements, cardiovascular health supplements and kids nutrition are the three biggest nutraceutical categories in China in terms of sales revenue, according to major e-commerce channel Alibaba Tmall.

Magtein, a branded form of magnesium L-threonate, has been approved as a novel food in the UK by Food Standards Agency (FSA) and has secured a five-year data protection period.

Dietary supplement brands selling on Amazon face a new compliance hurdle as the platform prepares to roll out an updated policy on March 31 that will increase scrutiny of product listings.

SB 2774 is part of a broader trend of state-level efforts to restrict youth access to certain dietary supplements, with similar proposals seen in over a dozen other states nationwide.

The European Union is set to ban monacolins from red yeast rice (RYR), a key ingredient in high cholesterol management, as early as Q3 2026, leaving the nutraceutical sector scrambling to fill the resulting ‘therapeutic gap’.

Professor Tim Spector says the Advertising Standards Authority (ASA) decision to uphold its ruling that Zoe’s Daily30+ supplement advertisement was misleading is a profound failure of logic.

A new survey covering global pharmacovigilance approaches in the natural health products sector is launching soon and will be extended to over 200 industry organizations and regulatory bodies, including those in China, the United States, Europe and other...

From state-level sales restrictions to federal preemption efforts and mounting questions around emerging ingredients, the dietary supplement industry is facing one of its most complex policy moments.

Experts have warned the future for berberine containing plants looks bleak after EFSA published a draft opinion questioning their safety, with a stakeholder consultation now open until May. 4.

Wild Nutrition’s registered trademark process ‘Food-Grown’ is deemed misleading to consumers by the Advertising Standards Authority (ASA).

With moves to close the self-GRAS “loophole”, plans to finally finalize the New Dietary Ingredient (NDI) guidance and talk of modernizing dietary supplement regulations, 2026 could be the biggest year since GMPs were introduced, says industry lawyer Ivan...

As regulatory enforcement and consumer awareness around dietary supplements continue to grow, manufacturers are under increasing pressure to demonstrate safety. At NOW Foods, that commitment includes employing a dedicated toxicologist, a role that...

Europe is taking a cautious approach as it reassesses how probiotics and the microbiome should be addressed within food and health regulation, attendees heard at the recent IPA World Congress + IPA 2026 event.

The Health Science Authority (HSA), which regulates health supplements in Singapore, is urging stronger collaboration between industry and academia to tackle the unique complexities of the booming longevity supplement sector.

Dsm-firmenich and Mara Renewables Corporation have been denied their appeals in relation to a recent UK High Court battle over three claims of patent infringement.

Dietary supplements are set to be formally recognized as a distinct category in global trade for the first time, following a recent update to the Harmonized System by the World Customs Organization.

The US imposing antidumping and countervailing duties on hard empty capsules from Brazil, China, India and Vietnam, China’s approval of 3’-sialyllactose (3’-SL) in infant formula are some of the latest updates in this regulatory news round-up.


The decision is likely to spark a wave of lawsuits from businesses across the food and beverage industry and beyond

The highly abundant South African polyphenol-rich marula fruit is being under-exploited by the functional food and supplements industry, according to the Marula Fruit Products Association.

EU packaging rules push supplement brands to show plans to cut excess plastic use by August 2026 under the new Packaging and Packaging Waste Regulation (PPWR).

With major evidence gaps remaining around how PFAS behave in soils and what this means for food systems, the government has launched new plan to track ‘forever chemicals’

A recent ruling could have dramatic implications on pricing, supply chains and the overall competitive landscape of Hard Empty Capsules.

Highly caffeinated energy pouches currently exist in a regulatory grey area, with a patchwork of different rules beginning to emerge across European nations.

An analysis of nearly 200 food supplements in Irish stores found a high prevalence of health claims but low compliance with the Nutrition and Health Claims Regulations (NHCR).

Progress Biotech’s DHA-algae oil will target infant nutrition as it receives organic certification in European first.

The Chinese authorities have approved 3’-sialyllactose (3’-SL) – a human milk oligosaccharide (HMO) – for use in infant formula for the first time.

FDA eases ‘no artificial colors’ claims, but ambiguity over ‘petroleum-based dyes’ leaves manufacturers cautious

A Republican congressman has introduced legislation seeking to reaffirm federal oversight of dietary supplements, blocking states from imposing their own additional regulatory requirements and creating what he characterized as a “confusing patchwork of...

Mouth pouches have emerged as one of the fastest growing CPG categories, signaling a major trend in nootropics as consumers seek functional ways to boost performance—sans nicotine.
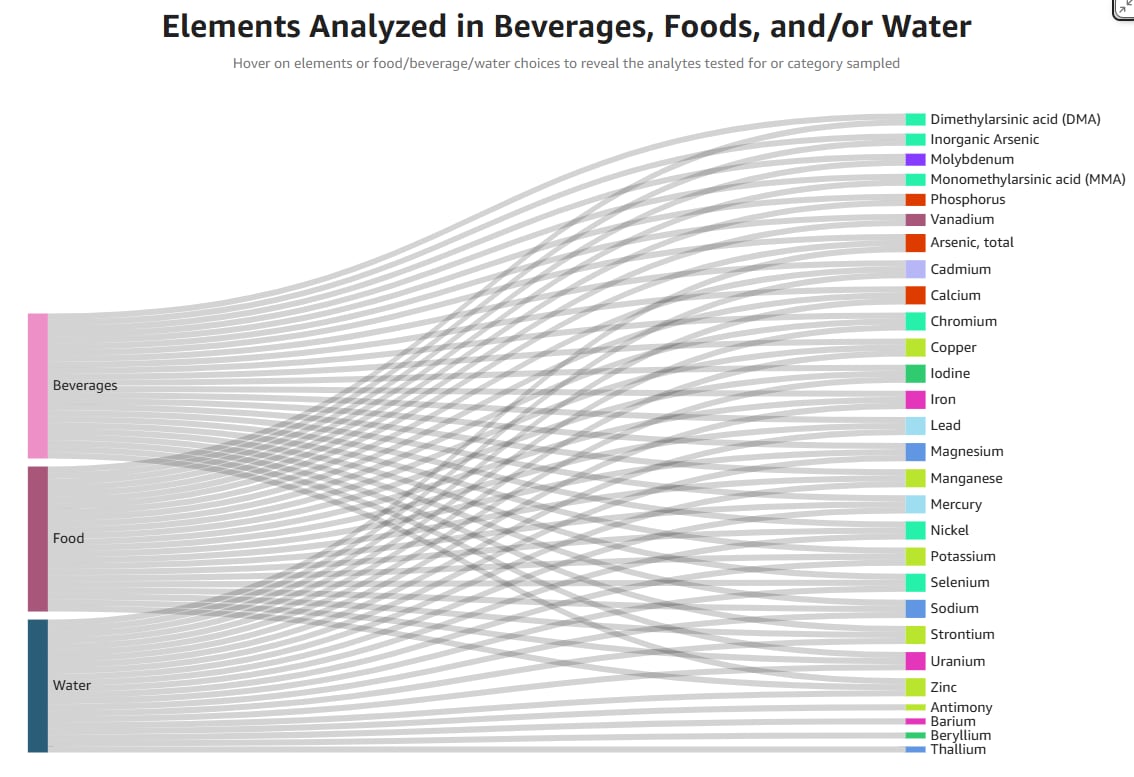
The FDA’s new Total Diet Study Interface is already being used to challenge state-level contamination claims, offering a clearer view of nutrient and contaminant data in the US food supply


Lawmakers in Hawaii have filed a bill to prohibit the sale of weight loss and muscle building products to minors, the latest in a long list of state-level attempts to restrict access to specific dietary supplement categories.

Ingredient distributor ACI Group has called on UK food producers to actively educate consumers about folic acid fortification, warning that misinformation about “ultra-processed foods” is overshadowing the health benefits of the policy.

South Korea’s Ministry of Food and Drug Safety (MFDS) is re-evaluating the safety and functionality of turmeric extract and eight other functional ingredients.

A scientific revolution is underway at the National Institutes of Health, emerging out of the ashes of the distrust instilled during the COVID-19 pandemic to reclaim science from the scientific aristocracy and make America healthy again.