Arizona State University researchers detail their findings, where they state most of the initial improvements in gut symptoms remained as well as improvements to the children’s’ language, behaviour and social interaction.
“We are finding a very strong connection between the microbes that live in our intestines and signals that travel to the brain," said Dr Rosa Krajmalnik-Brown, study team member and associate professor at the University. "Two years later, the children are doing even better, which is amazing."
"Many kids with autism have gastrointestinal problems, and some studies, including ours, have found that those children also have worse autism-related symptoms," she added.
"In many cases, when you are able to treat those gastrointestinal problems, their behaviour improves."
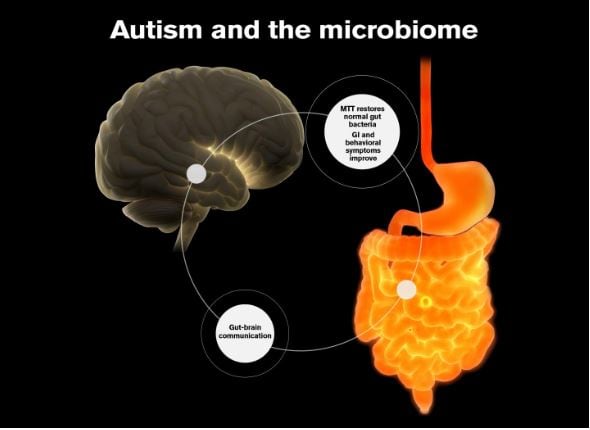
Individuals with Autism Spectrum Disorders (ASD), Parkinson’s disease, and Alzheimer’s disease, may experience chronic gastrointestinal (GI) symptoms suggesting the presence of a gut-brain axis.
Growing number of research groups have observed that children with ASD have distinctive gut microbiomes compared to neurotypical children.
While effective approaches for ASD include dietary and nutritional strategies, no approved medical treatment exists to treat ASD symptoms, such as social communication difficulties and repetitive behaviours.
Considering the link between the gut and brain, modulating the gut microbiome by antibiotics, probiotics, prebiotics, and/or faecal microbiota transplant (FMT) could be a viable therapeutic option.
The research team were following up on an earlier open-label trial carried out in 2017 that combined antibiotics, a bowel cleanse, a stomach-acid suppressant, and faecal microbiota transplant.
‘A long term impact’
At the time the study, led by Krajmalnik-Brown and Adams, concluded that "Improvements in GI symptoms, ASD symptoms, and the microbiome all persisted for at least eight weeks after treatment ended, suggesting a long-term impact."
In the current study, the same 18 children with autism spectrum disorder (ASD) were invited to participate in a follow-up, where a selection of gastrointestinal (GI) and behaviour tests were carried out.
Twelve of 18 participants made some changes to their medication, diet, or nutritional supplements, but these changes were well documented and were mostly minor.
The researchers found that after two years post-treatment, most of the initial improvements in gut symptoms remained.
In addition, parents reported a slow steady reduction of ASD symptoms during treatment and over the next two years.
Professional evaluation revealed a 45% decrease in ASD symptoms (language, social interaction and behaviour) compared to baseline.
Researchers also note that although there may be some placebo effect, much of that effect appears to be real.
At the start of the study, 83% of participants were rated as "severe" autism. At the end of the study, only 17% were "severe," 39% were "mild/moderate," and 44% were below the cut-off for mild ASD.
‘Slow but steady improvement’
“Understanding which microbes and chemicals produced by the microbes are driving these behavioural changes is at the heart of our work," Krajmalnik-Brown said.
“The team's new publication reports that the study demonstrated that two years after treatment stopped the participants still had an average of a 58% reduction in GI symptoms compared to baseline.
“In addition, the parents of most participants reported "a slow but steady improvement in core ASD symptoms."”
"Drs. Krajmalnik-Brown, Kang and I are excited about the results, but we want to caution the public that we need larger clinical trials for this to become an FDA-approved treatment," said co-author Dr James Adams. “Professional expertise is required for safe and effective treatment.”
He also pointed out that some participants in the trial shared common traits, including birth by C-section, reduced breastfeeding, increased antibiotics, and low fibre intake by the mother and child.
Dr Adams said these traits can lead to limited biodiversity in their gut bacteria adding that the open label nature of the study and the small sample size used, more research was needed in order to verify the usefulness of MTT as a therapeutic.
“These are encouraging results, but this is very early-stage research involving just a small sample of children,” agreed the NHS website, the UK's biggest health online resource.
“Though no adverse effects were reported, the safety of the procedure would need to be determined in a much larger trial.
“There's also the possibility that the results were due to the placebo effect, so a randomised controlled trial is needed to make sure that the treatment works.”
Source: Scientific Reports
Published online: doi.org/10.1038/s41598-019-42183-0
“Long-term benefit of Microbiota Transfer Therapy in Autism Symptoms and Gut Microbiota."
Authors: Dae-Wook Kang, Rosa Krajmalnik-Brown, James Adams