Gut microbes may provide some protection against radiation-based treatments: Study
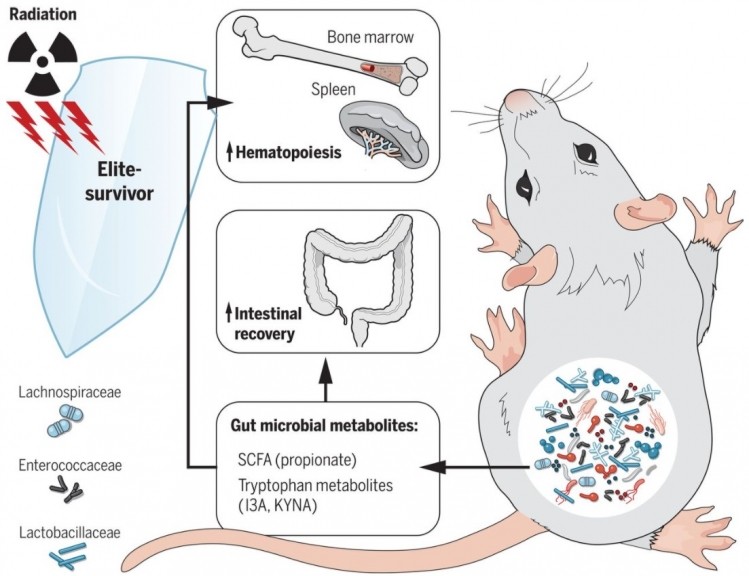
Writing in Science, the team identifies Lachnospiraceae and Enterococcaceae as bacteria possibly responsible, supported by studies that found high numbers of these species in leukaemia patients, who underwent radiotherapy.
“Substantial federal efforts have been made to mitigate acute radiation symptoms — however, it remains a long-standing and unresolved problem,” says first study author Hao Guo, a postdoctoral fellow.
“Our work produced a comprehensive dataset of bacteria and metabolites that can serve as a powerful resource to identify actionable therapeutic targets in future microbiome studies.”
Previous findings have pointed to possibilities that the gut microbiota and metabolites play a key role in the regulation of disease susceptibility after radiation treatments.
Gut microbes, as well as microbe-derived metabolites, such as short-chain fatty acids (SCFAs) and tryptophan metabolites, all play a role in regulating host metabolism and immunity.
The imbalance of the gut’s microbial community is linked to disease incidence, risks and even the clear onset of clinical symptoms.
The research team from University of North Carolina at Chapel Hill have previously highlighted the importance of gut microbiota, the Lachnospiraceae bacteria and SCFAs in reducing colitis and obesity.
It has also been reported that SCFAs and tryptophan metabolites can reduce proinflammatory cytokines and promote anti-inflammatory cytokines, which are all mediators of radiation-induced damage.
Lethal radiation levels
Along with colleagues from Duke University, Memorial Sloan Kettering and Weill Cornell Medical College, the team subjected mice to potentially lethal levels of total body radiation.
They found the presence of Lachnospiraceae and Enterococcaceae, which together led to increased production of propionate and tryptophan.
These metabolites play a role in protection from radiation, lessened damage to bone marrow stem cell production and development of gastrointestinal problems as well as reduce DNA damage.
Both metabolites can be purchased in some countries as health supplements but there is currently no evidence that the supplements could aid people exposed to intense forms of radiation.
“Granulocyte-colony stimulating factor is the only drug that has been approved by the FDA as an effective countermeasure for high-dose radiation exposure, but it is expensive and has potential adverse side-effects,” explains corresponding author Jenny Ting, William Rand Kenan Professor of Genetics at the UNC School of Medicine.
“However, bacteria that we can cultivate, and especially metabolites that are relatively inexpensive and already elements in the food we eat, may be a good alternative.”
Leukaemia patients
Along with colleagues from Duke University, Memorial Sloan Kettering and Weill Cornell Medical College, the team then took a look at faecal samples from 21 leukaemia patients due to receive radiation therapy.
The scientists found that patients with shorter periods of diarrhoea had significantly higher abundances of Lachnospiraceae and Enterococcaceae than patients with longer periods of diarrhoea.
These findings are similar to the researchers’ findings in mice although Ting advises larger studies are needed to confirm these outcomes.
“Our findings emphasise a crucial role for the gut microbiota as a master regulator of host defence against radiation, capable of protecting both the hematopoietic and gastrointestinal systems,” the paper concludes.
“Lachnospiraceae and Enterococcaceae, together with downstream metabolites represented by propionate and tryptophan pathway members, contribute substantially to radioprotection.
“This study sheds light on the pivotal role that the microbiota-metabolite axis plays in generating broad protection against radiation and provides promising therapeutic targets to treat the adverse side effects of radiation exposure.”
Source: Science
Published online: DOI: 10.1126/science.aay9097
“Multi-omics analyses of radiation survivors identify radioprotective microbes and metabolites.”
Authors: Hao Guo et al.