The research centers around horizontal gene transfer (HGT), a process in which individual cells mix their genomes and acquire new functions from other microbes, which happens without having to reproduce or be inherited. According to Mathilde Poyet, PhD, co-author of the study, HGT is key in understanding how bacteria evolve within us and how our daily activities and lifestyle choice may influence the composition of their genomes.
Previous research has highlighted how the composition of the microbiome of people living in industrialized societies differs from that of rural individuals living in relative isolation. Non-industrialized populations usually have a larger bacterial biodiversity, including many species that are not seen in industrialized populations. Differences in diet, antibiotic use, and exposure to soil bacteria are thought to be contributing factors.
The research, published in Cell, is the first major paper from the Global Microbiome Conservancy (GMbC), a nonprofit organization that works with researchers all around the world to preserve bacterial species that are at risk of being lost as humanity becomes more exposed to industrialized diets and lifestyles globally.
Data
The research team cultured, isolated, and sequenced over 4,000 bacterial strains from stool samples of 37 people in 14 populations, including the USA, Canada, Finland, Cameroon, Tanzania, Ghana, and Nigeria.
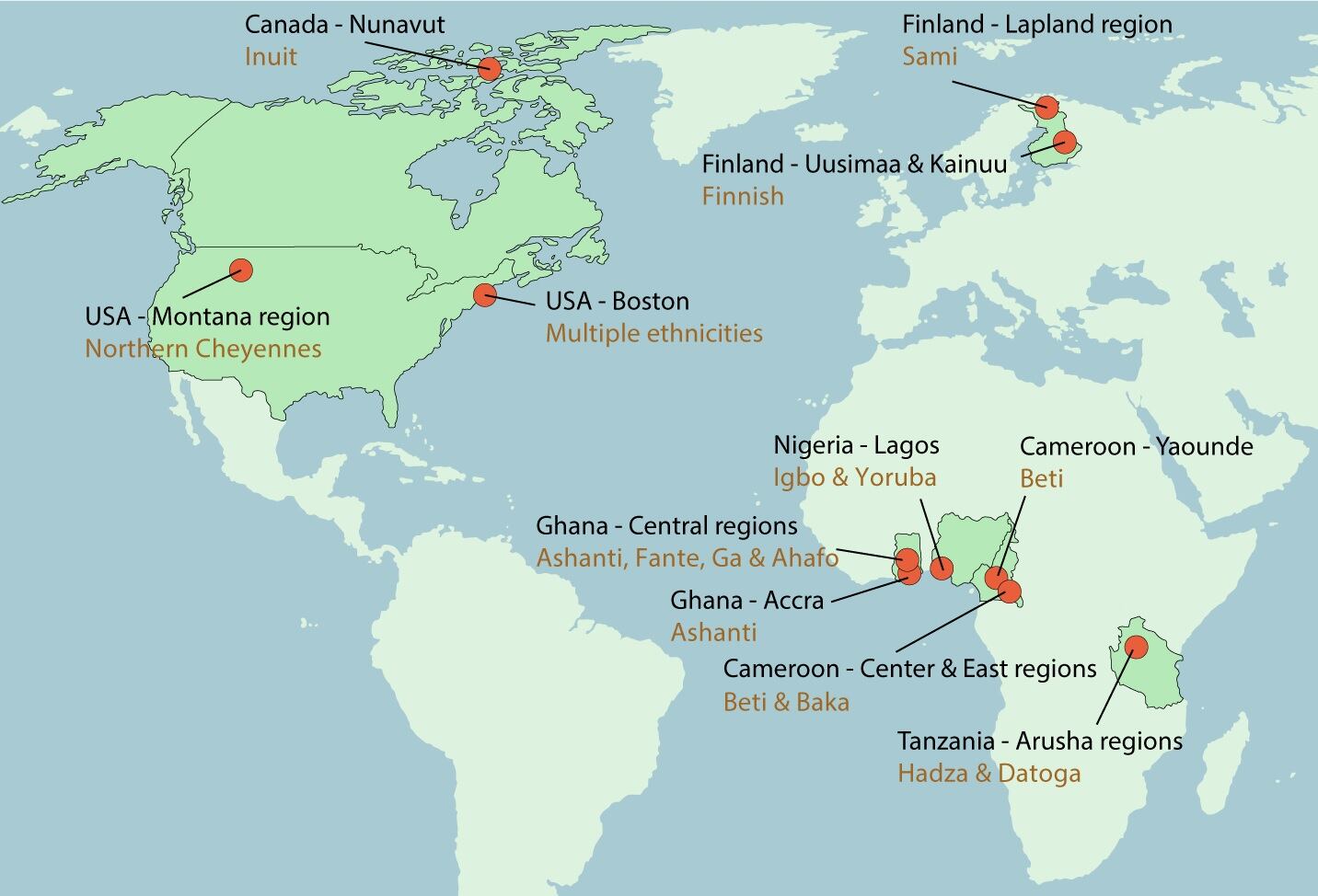
The researchers combined this dataset with data from 11 people living in the northeastern US, and then scored each population as industrialized or not (defined by the United Nation’s Human Development Index) and urban or rural (measured by population density).
Participants represented four different lifestyle categories: urban industrialized (UI) individuals in the USA and eastern and southern Finland; rural industrialized (RI) individuals in the USA, arctic Finland, and the Canadian arctic; urban non-industrialized (UN) individuals in Cameroon, Nigeria, and Ghana; and rural non-industrialized (RN) individuals in Cameroon, Tanzania, and Ghana.
Findings
Depending on the species, the researchers found that bacteria might obtain between 10 and 100 new genes every year. It was also discovered that the rate of gene exchange was significantly higher in people living in industrialized societies, and they also observed differences in the types of genes that were most commonly exchanged.
Poyet told NutraIngredients-USA that there are three major takeaways from the study:
1. First, the results show that gene transfers between human gut bacterial species occur frequently within the gut of each person, illustrating how bacterial genomes become personalized to their human host.
2. Second, by comparing HGTs across human populations we revealed that industrialized lifestyles are linked with an increased frequency of gene transfers in the human gut microbiome.
3. Finally, the data the team generated also allowed them to discover that the functions of these HGTs reflect the host’s lifestyle and are related to their level of industrialization.
“For example, non-industrialized cohorts, who consume larger amounts of non-digestible fiber, harbored gut bacteria that exchanged the enzyme required to digest these fibers (CAZyme) genes at higher frequencies than individuals living in industrialized and/or urban regions. Another example concerns gut bacteria of pastoralists in Tanzania, who frequently administer antibiotics to their herds, and whose gut bacteria exchange great amounts of antibiotic resistance genes,” explained Poyet.
Implications
Poyet explained that this research gives perspective on the consequences that industrialized lifestyles has on our microbiome. “It also furthers our understanding of how bacteria adapt in the gut environment. In the long term, it will help with predicting the consequence of treatments such as Fecal Material Transplants (FMTs) or probiotic intake on the dynamics of gene exchange between bacteria upon transfer into recipient patients.”
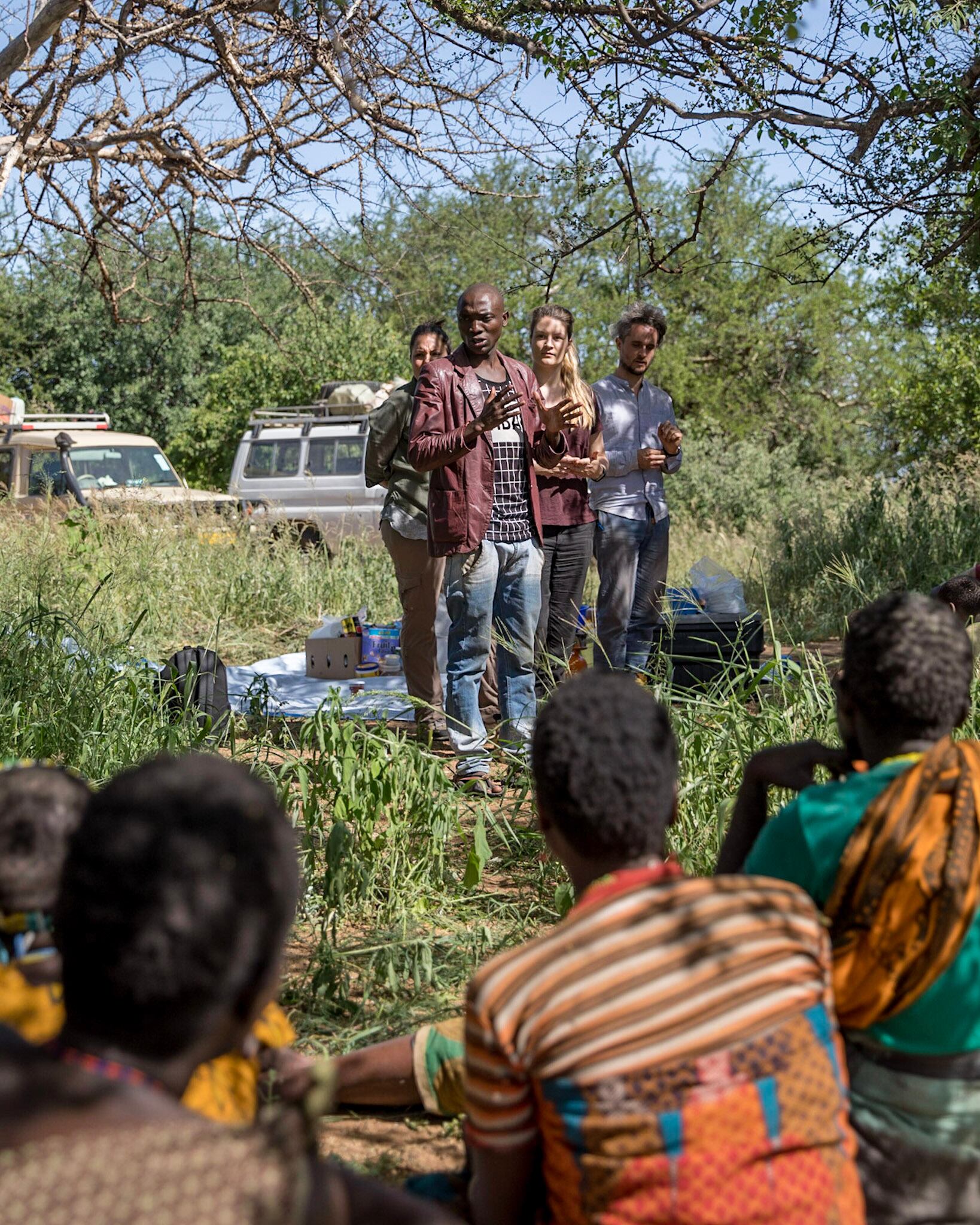
Poyet, who co-founded GMbC with co-author Mathieu Groussin in 2016, said that collecting microbiome samples from underrepresented human populations is a key part of their mission.
“GMbC is committed to advancing equity through science, by: prioritizing groups who have been historically underrepresented in biomedical research; partnering with local researchers in the regions where we work, and supporting and amplifying their research; and implementing a unique model for material and IP ownership that directly shares benefits with participating communities,” explained Poyet. “By collecting and biobanking this important biodiversity, we aim to build a resource for scientists around the world to advance microbiome research and human health.”
Source: Cell
31 March 2021 doi.org/10.1016/j.cell.2021.02.052
“Elevated rates of horizontal gene transfer in the industrialized human microbiome”
Authors: M. Groussin et al.