AB-Biotic probiotic blend shows efficacy for infant GI disorders
Published in Frontiers in Microbiology, the research findings point to AB Biotics’ B. longum KABP042 and P. pentosaceus KABP041 strains as effective against disorders such as colic and constipation.
Scientists from the Spain-based pharma firm and the University of Catalonia present key findings that include the strains’ ability to adhere to human intestinal epithelium.
The team also noted the blend’s protective qualities of the intestinal barrier, achieved by regulating the expression of tight junction proteins.
Both strains also demonstrated broad pathogen inhibition capacity via the production of organic acids and antimicrobials.
Methodical approach
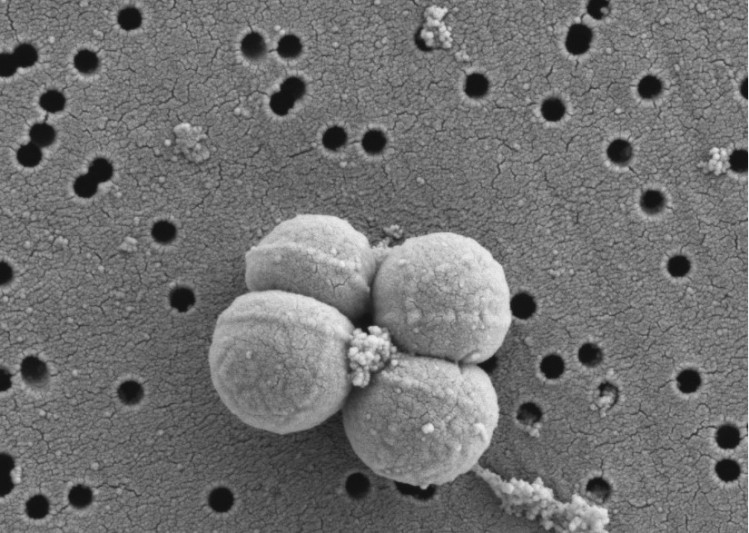
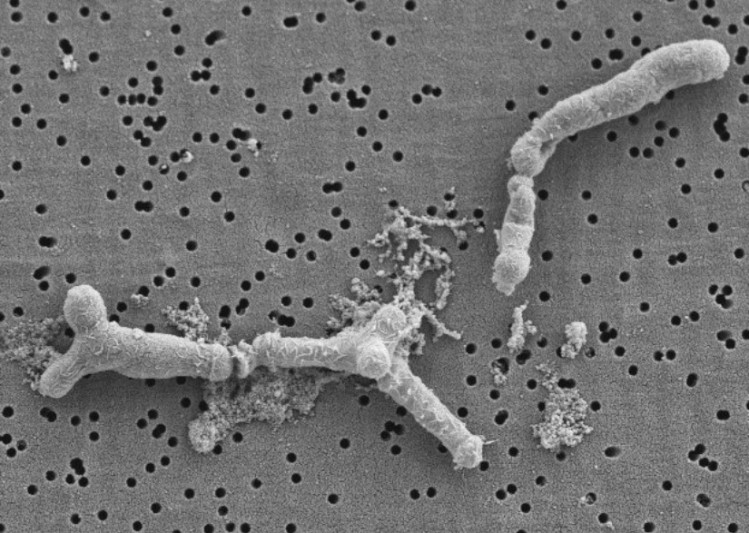
The team began sequencing the genome of both strains, also adopting in silico analyses, where no virulence factors were detected for any strain.
Only the non-transferable erm(49) gene, which confers resistance to erythromycin and clindamycin, was identified in the genome of B. longum KABP042.
Further analysis via in vitro characterisation revealed the strains’ tolerance of gastric and bile challenges and strong ability to ‘stick’ to human intestinal cells.
The researchers suggest the two strains combine to induce the expression of Caco-2 tight junction proteins, which are essential for the maintenance of barrier integrity.
Additional growth inhibition experiments by the team demonstrated the ability of the two strains alone and in combination to antagonise Gram-negative and Gram-positive bacterial pathogens.
“Pathogens’ inhibition was mostly mediated by the production of organic acids,” the researchers write.
“But neutralisation experiments strongly suggested the presence of additional antimicrobial compounds in probiotic culture supernatants such as the bacteriocin Lantibiotic B.”
Pilot study
Finally, an exploratory, observational, pilot study involving 36 infants diagnosed with at least one FGID (infant colic and/or functional constipation) showed the probiotic formula was well tolerated.
FGID severity was significantly reduced after 14 days of treatment with the two strains as the team concluded that the study: “confirms the strains fulfil probiotic requisites and show functionality over intestinal barrier integrity and pathogen inhibition.”
“In addition, the outcome of the observational, single-arm, pilot trial confirms the safety of the probiotic formula and set the basis for future larger, randomised, double blinded, placebo-controlled clinical trials in infants with diverse FGIDs.”
Source: Front. Microbiol
Published online: doi.org/10.3389/fmicb.2021.741391
“Probiotic Properties of Bifidobacterium longum KABP042 and Pediococcus pentosaceus KABP041 Show Potential to Counteract Functional Gastrointestinal Disorders in an Observational Pilot Trial in Infants.”
Authors: Erola Astó et al.