Promotional Features
How a potent new combination of herbal actives is offering a new approach to inflammation control
Inflammation is linked to a wide range of diseases, including arthritis, cancer, cardiovascular disease, diabetes, obesity, osteoporosis, inflammatory bowel disease and asthma. It can even be a factor in the development of diseases of the central nervous system such as Parkinson's, and mental health disorders such as depression. [1]
Research suggests that almost 90% of synthetic anti-inflammatory drugs can cause drug-related toxicities and additional adverse effects. [2, 3] Consequently, there has been a resurgence in interest in herbal ingredients with anti-inflammatory properties. The most promising and widely used herbs in this context are Curcuma longa and Boswellia serrata, both of which command high levels of consumer awareness and market demand.
CurcuminCurcuma, a compound which has been used in traditional Ayurvedic medicine for thousands of years, is well known for its antioxidant and anti-inflammatory properties. [4-7] The three major curcuminoids isolated from Curcuma longa are curcumin, demethoxycurcumin and bisdemethoxycurcumin, with curcumin being the primary component. Numerous studies have demonstrated its biological activity and it has been used to promote joint, brain, cardiovascular and gastro intestinal health.
Curcumin targets multiple molecular targets and mechanisms of action that are linked to anti-inflammatory activity. It mainly affects and modulates the presence of proinflammatory signaling molecules and enzymes by down-regulating or inhibiting pro-inflammatory mediators and cytokines. [7,8]
Growing awareness of these biological properties has resulted in a significant increase in the number of health and wellness products containing curcumin. Indeed, it has become one of the top selling herbal ingredients in Europe and the US.
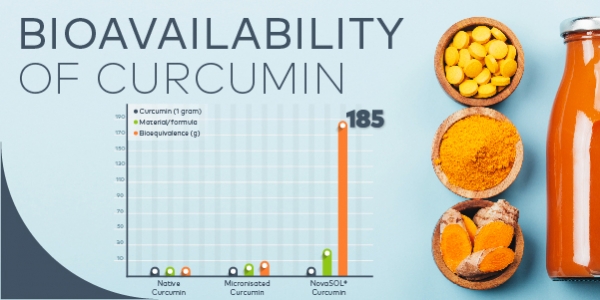
However, the low bioavailability of curcumin is a major hurdle [9] and various techniques have been developed to overcome this limitation. The challenge is to achieve biologically relevant blood serum levels of curcumin, and many formulators find they are not able to accommodate relevant dosages, thus limiting the bioactivity of the finished products.
Now there is a new solution. NovaSOL is a proprietary technology based on micellar solubilization of herbal extracts. This results in a liquid formula with biomimetic (“nature-like”) properties which enhance the solubility and gastrointestinal absorption of orally administered herbal actives. Micelle encapsulation allows for the use of smaller active ingredient dosages in finished products that result in relevant biological activity.
The NovaSOL micellation of curcumin has been shown to result in the largest increase of curcumin bioavailability reported to date. [10-12] In a human clinical trial, a 185-fold larger area under the curve (AUC) and a 455-fold higher maximum total curcumin plasma concentration (Cmax) were observed for micellar curcumin as compared to dosing with the native form. [12]
NovaSOL Curcumin offers a liquid, soluble form of curcumin extract which results in significant uptake of curcuminoids even while using a relatively low daily dose.
BoswelliaIn recent years, the use of Boswellia serrata extracts as a natural, herbal anti-inflammatory ingredient has gained popularity. The number of scientific publications describing its biological activity also has increased substantially; Boswellia extracts have been shown to have a range of positive effects on health, mainly in joint, gastrointestinal and respiratory health. [13-18]
The mechanism of the anti-inflammatory action of Boswellia extracts stems from the boswellic acids (BAs) which are the active principals of frankincense. They have been shown to inhibit a number of enzymes that are pivotal to the inflammatory process, as well as several pro-inflammatory cytokines. One specific compound, AKBA (3-O-acetyl-11-keto-boswellic acid), has been shown to modulate specific inflammatory pathways and is the most active BA. [19-21]
Like Curcuma extracts, Boswellia extracts usually have poor bioavailability because of their hydrophobicity and poor water solubility. Pharmacokinetic studies conducted in human and animal models have indicated that after oral administration of Boswellia products, a sufficient systemic concentration of AKBA is required in order to achieve anti-inflammatory effects in vivo. [22–26] Poor absorption through the intestine affects the systemic availability of AKBA, thus limiting the anti-inflammatory activity of standard Boswellia extracts.
Bioavailability can be significantly increased through the preparation of a micellar formula in the form of liquid NovaSOL Boswellia. The administration of a Boswellia extract in its soluble form has been shown to result in a significant increase of BA plasma concentrations. This is particularly true for AKBA, the bioavailability of which is increased up to 50-fold with micellar formulation. [27]
The value of combining curcumin and BoswelliaThe activity of curcumin and Boswellia extracts individually has been well defined – and combining them is expected to promote their multi-target biological activity on inflammatory pathways. The diversity in the mechanism of action of these actives is expected to result in a complementary effect with additional positive health effects. Micellar solubilization of the combination of extracts (curcumin and Boswellia) also should result in a higher level of anti-inflammatory activity compared to their respective native forms.
A recent study has shown that combining liquid, micellar curcumin with Boswellia not only increased bioavailability, but also resulted in enhanced biological activity. [28] The combination was found to result in a significantly more potent anti-inflammatory effect as compared to effects seen with native and standalone extracts. Additional research is ongoing to further investigate the activity of the combined formula.
NovaSOL Curcumin / Boswellia offers a potent, complementary formula of natural actives with high bioavailability. Its use is based in science and has been formulated to deliver optimal biological activity in a convenient dose form.
References
[1] Laveti, D., Kumar, M., Hemalatha, R., Sistla, R., et al. Anti-inflammatory treatments for chronic diseases: a review. Inflamm. Allergy Drug Targets, 2013, 12, 349-361
[2]Lanas, A. Non-steroidal anti-inflammatory drugs and cyclooxgenase inhibition in the gastrointestinal tract: a trip from peptic ulcer to colon cancer. Am. J. Med. Sci. 2009, 338, 96- 106.
[3] Kandulski, A., Venerito, M., Malfertheiner, P. Non-steroidal anti-inflammatory drugs (NSAIDs) – balancing gastrointestinal complications and the cardiovascular risk. Dtsch. Med. Wochenschr. 2009, 134, 1635-1640.
[4] Prasad, S., Gupta, S.C., Tyagi, A.K., Aggarwal, B.B. Curcumin, a component of golden spice: from bedside to bench and back. Biotechnol Adv. 2014, 32, 1053-1064
[5] Amalraj, A., Varma, K., Jacob, J., Divya, C., Kunnumakkara, A.B., Stohs, S.J., Gopi, S. A Novel Highly Bioavailable Curcumin Formulation Improves Symptoms and Diagnostic Indicators in Rheumatoid Arthritis Patients: A Randomized, Double-Blind, Placebo- Controlled, Two-Dose, Three-Arm, and Parallel-Group Study. J. Med. Food., 2017, 20, 1022-1030
[6] Moghadamtousi, S.Z., Kadir, H.A., Hassandarvish, P., Tajik, H., Abubakar, S., Zandi, K. A review on antibacterial, antiviral, and antifungal activity of curcumin. Biomed. Res. Int., 2014, ID186864
[7] Jurenka, J.S. Anti-inflammatory properties of curcumin, a major constituent of Curcuma longa: A review of preclinical and clinical research. Altern. Med. Rev. 2009, 14, 141–153
[8] Bisht, K., Wagner, K.H., Bulmer, A.C. Curcumin, resveratrol and flavonoids as antiinflammatory, cyto- and DNA-protective dietary compounds. Toxicol. 2010, 278, 88–100.
[9] Douglass, B.J., Clouatre, D.L. Beyond yellow curry: Assessing commercial curcumin absorption technologies. J. Am. Coll. Nutr. 2015, 34, 347–358
[10] Schiborr, C., Kocher, A., Behnam, D., Jandasek, J., et al. The oral bioavailability of curcumin from micronized powder and liquid micelles is significantly increased in healthy humans and differs between sexes. Mol. Nutr. Food Res., 2014, 58, 516–527
[11] Kocher, A., Schiborr, C., Behnam, D., Frank, J. The oral bioavailability of curcuminoids in healthy humans is markedly enhanced by micellar solubilisation but not further improved by simultaneous ingestion of sesamin, ferulic acid, naringenin and xanthohumol. J. Funct Foods 2015,14, 183–191
[12] Kocher, A., Bohnert, L., Schiborr, C., Frank, J. Highly bioavailable micellar curcuminoids accumulate in blood, are safe and do not reduce blood lipids and inflammation markers in moderately hyperlipidemic individuals. Mol. Nutr. Food Res. 2016, 60, 1555-1563
[13] Sander O, Herborn G and Rau R: Is H15 (resin extract of Boswellia serrata, ‘incense’) a useful supplement to establish drug therapy of chronic polyarthritis? Results of a double-blind pilot study. Z Rheumatol 57: 11-16, 1998 (In German)
[14] Gerhardt H, Seifert F, Buvari P, et al: Therapy of active Crohn disease with Boswellia serrata extract H15. Z Gastroenterol 39: 11-17, 2001 (In German)
[15] Kimmatkar N, Thawani V, Hingorani L and Khiyani R: Efficacy and tolerability of Boswellia serrata extract in treatment of osteoarthritis of knee - a randomized, double-blind, placebo-controlled trial. Phytomedicine 10: 3-7, 2003
[16] Sontakke S, Thawani V, Pimpalkhute P, et al: Open, randomized, controlled clinical trial of Boswellia serrata extract as compared to valdecoxib in osteoarthritis of the knee. Indian J Pharmacol 39: 27-29, 2007
[17] Sengupta K, Alluri KV, Satish AR, et al: A double-blind, randomized, placebo-controlled study of the efficacy and safety of 5-Loxin for treatment of osteoarthritis of the knee. Arthritis Res Ther 10: R85, 2008
[18] Madisch A, Miehlke S, Eichele O, et al: Boswellia serrata extract for the treatment of collagenous colitis. A double-blind, randomized, placebo-controlled, multicenter trial. Int J Colorectal Dis 22: 1445-1451, 2007
[19] Safayhi H, Mack T, Sabieraj J, Anazodo MI, Subramanian LR and Ammon HP: Boswellic acids: novel, specific, nonredox inhibitors of 5-lipoxygenase. J Pharmacol Exp Ther 261: 1143-1146, 1992
[20] Sailer ER, Subramanian LR, Rall B, Hoernlein RF, Ammon HP and Safayhi H: Acetyl-11-keto-beta-boswellic acid (AKBA): structure requirements for binding and 5-lipoxygenase inhibitory activity. Br J Pharmacol 117: 615-618, 1996
[21] Sailer ER, Schweizer S, Boden SE, Ammon HP and Safayhi H: Characterization of an acetyl-11-keto-beta-boswellic acid and arachidonate-binding regulatory site of 5-lipoxygenase using photoaffinity labeling. Eur J Biochem 256: 364-368, 1998
[22] Abdel Tawab M, Kaunzinger A, Bahr U, Karas M, Wurglics M, Schubert-Zsilavecz M (2001) Development of a high-performance liquid chromatographic method for the determination of 11-keto-b-boswellic acid in human plasma. J Chromatogr Biomed Appl 761:221–227
[23] Buchele B, Simmet T (2003) Analysis of 12 different pentacyclic triterpenic acids from frankincense in human plasma by high-performance liquid chromatography and photodiode array detection. J Chromatogr B 795:355–362
[24] Sharma S, Thawani V, Hingorani L, Shrivastava M, Bhate VR, Khiyani R (2004) Pharmacokinetic study of 11-keto-b-boswellic acid. Phytomedicine 11:1255–1260
[25] Sterk V, Buchele B, Simmet T (2004) Effect of food intake on the bioavailability of boswellic acids from a herbal preparation in healthy volunteers. Planta Med 70:1155–1160
[26] Kruger P, Daneshfar R, Eckert GP, Klein J, Volmer DA, Bahr U, Muller WE, Karas M, Schubert-Zsilavecz M, Abdel Tawab M (2008) Metabolism of boswellic acids in vitro and in vivo. Drug Metab Depos 36:1135–1142
[27] Mona Abdel-Tawab. Enhanced absorption of boswellic acids by a micellar solubilized delivery form of Boswellia extract. NFS Journal 11:12-16, 2018.
[28] T. Khayyal et al. Micellar solubilisation enhances the anti-inflammatory activities of curcumin and boswellic acids in rats with adjuvant-induced arthritis. Nutrition 54:189-196, 2018.