Celiac disease is a chronic autoimmune disease that affects the small intestine in response to the ingestion of gluten, a protein found in wheat, barley and rye, and affects about 1% of the global population.
In people with celiac disease, the immune system mistakenly attacks the lining of the small intestine when gluten-containing foods are ingested, which can lead to damage and inflammation of the intestinal wall (Figure 1). This mechanism causes the alteration of the intestinal mucosa, characterized by atrophy of the villi (flattening) with excessive accumulation of pro-inflammatory cells, thus limiting the normal function of nutrient absorption in the intestinal tract.
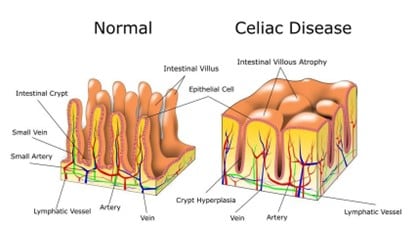
Above: Representation of normal and celiac Intestinal Villi
Symptoms of celiac disease can vary widely from person to person and may include abdominal pain, bloating, diarrhoea, constipation, fatigue, anaemia, weight loss, and rash. Celiac disease, if not treated, can lead to complications such as malnutrition, osteoporosis, and an increased risk of certain types of cancer.1
The only treatment for celiac disease is a strict, lifelong gluten-free diet; this involves eliminating all foods and products that contain this protein, including bread, pasta, cereals and many processed foods. In some cases, supplements or medications may also be recommended to help manage symptoms or address nutritional deficiencies.
The diagnosis of celiac disease is made through a blood sample to search for specific antibodies to gluten and then a biopsy of the small intestine to look for signs or damage to the intestinal wall, typical of celiac disease.2
Iron deficiency anaemia (IDA) is a common complication in celiac patients. National and international guidelines recommend screening for the evaluation of iron parameters in this category of subjects, such as haemoglobin and specific parameters of iron metabolism that indicate the amount of iron present in the body (serum iron and ferritinaemia).
Iron deficiency anemia
According to the World Health Organization (WHO) the physiological range of haemoglobin (Hb) varies according to gender and age: men 14-18 g/dL, women 12-16 g/dL and children 10.5-13.5 g/dL.
When the Hb value is lower than the above values, we speak of iron deficiency anaemia, which is also confirmed by the measurement of other important parameters: Ferritin, storage protein capable of containing up to 4500 iron atoms (physiological range: 30-300 ng/mL);
- Sideremia, which indicates the amount of circulating iron bound to a specific protein, called transferrin (physiological range: 50-180 mcg/dL)
- Transferrin Saturation (%TSAT), a protein that transports iron in the tissues and districts responsible for its use (physiological value: >20%)
When it comes to sideropenia, however, there are not always evident haematological alterations; in fact, haemoglobin levels may be normal, but ferritin and transferrin saturation are low. In this case it is strongly recommended to increase the amount of iron through diet or food supplementation.
The incidence of iron deficiency is found in 10-15% of subjects suffering from celiac disease.4
Iron and celiac disease supplementation
Iron supplementation is often required in patients with celiac disease who can have poor iron absorption due to intestinal inflammation.3 Oral iron formulations are often preferred over intravenous formulations, due to the greater convenience of use and patient preferences. However, supplementation of the standard oral iron formulations in celiac patients may aggravate some symptoms of the disorder, such as abdominal pain, bloating, diarrhea and constipation.5
Sucrosomial® iron – clinical evidence
To counteract the states of iron deficiency, PharmaNutra Spa has developed Sucrosomial® Iron, marketed under the name of Sideral®.
This new technology represents an innovative oral formulation containing iron, in which iron pyrophosphate is wrapped in a matrix of phospholipids and sucrose esters of fatty acids; this structure is called Sucrosome®.4
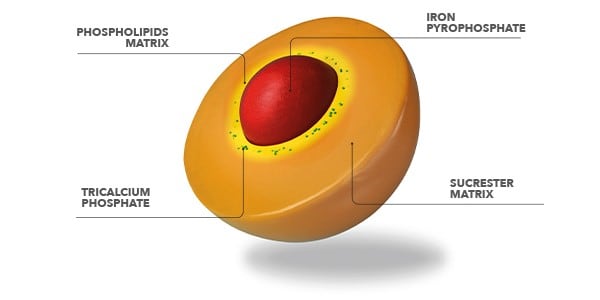
Above: Structure of the Sucrosome® containing iron pyrophosphate
The result of this technology leads to a high gastrointestinal tolerability and easier absorption of iron in the intestine. In recent years, many scientific evidences (both preclinical and clinical) have been published in international scientific journals confirming the effectiveness of Sucrosomial® technology.
Gastro resistance is given by the sucrester matrix, as demonstrated in in-vitro studies, which protects iron from the acidity of the pH of the stomach.
This allows the Sucrosome® to arrive intact in the intestinal mucosa, where it is absorbed by enterocytes as a vesicular structure. This is a completely different mode of absorption compared to conventional iron which is absorbed by a specific membrane channel placed on the apical part of the enterocytes, called Divalent Metal Transporter type 1 (DMT1), responsible for the reduced absorption of iron and, consequently, its common adverse effects (e.g. gastralgia).5
In a clinical study published in 2018, 43 subjects with celiac disease were divided into two groups: the first consisted of 24 subjects, who had reported previous side effects following the use of iron sulphate, were supplemented with 30 mg of SiderAL® for 90 days; the second group (control) consisted of 19 subjects, who were tolerant to iron sulphate, and continued the same treatment, at a dosage of 105 mg per day, for 90 days. 6
The results showed that treatment with SiderAL® increased haemoglobin values by administering a dosage three times lower than iron sulphate.
In addition, subjects taking Sucrosomial® iron experienced less severe gastrointestinal symptoms and improved overall wellbeing and, in contrast to the control group treated with iron sulphate, which experienced a greater severity of gastrointestinal symptoms during the study period.
In conclusion, Sucrosomial® iron can be better tolerated and is suitable for patients suffering from celiac disease and iron deficiency anaemia, since its innovative formulation allows good intestinal absorption even when the intestinal mucosa is inflamed.6

References
1. Mahadev S., et al. Prevalence of Celiac Disease in Patients with Iron Deficiency Anaemia – a Systematic Review with Meta-analysis. Gastroenterology. 2018, 155(2): 374-382.
2. Marsh, M.N. The small intestine: Mechanism of local immunity and gluten sensitivity. Clinical Science 1981, 61, 497-503.
3. Malterre T. Digestive and nutritional considerations in celiac disease: Could supplementation help? Altern. Med. Rev. 2009;14:247–257.
4. Gómez-Ramírez S., et al.; Sucrosomial® Iron: A New Generation Iron for Improving Oral Supplementation. Pharmaceuticals (Basel). 2018;11(4):97.
5. Elli L., et al. Nomenclature and diagnosis of gluten-related disorders: A position statement by the Italian Association of Hospital Gastroenterologists and Endoscopists (AIGO) Dig. Liver Dis. 2017;49:138–146.
6. Elli L., et al. Sucrosomial Iron Supplementation in Anemic Patients with Celiac Disease Not Tolerating Oral Ferrous Sulfate: A Prospective Study. Nutrients. 2018;10(3):330.





