Promotional Features
MAKING MOVES IN JOINT HEALTH WITH NATIVE TYPE II COLLAGEN
Collagen is becoming increasingly popular in the joint health market. Discover the benefits of native type II collagen for convenient and low-dose supplements.
Widely used in beauty and cosmetics, as well as food and beverage applications, collagen is now making big moves in the joint health market, creating exciting opportunities for dietary supplement manufacturers to innovate in this sector.
Collagen is the main component of connective tissues that make up tendons, ligaments, skin and cartilage. Although it has many important functions in the body, collagen is best known for its structural role – providing a framework for tissues throughout the body.[1] Of the 28 different types of collagen that have been identified, type II collagen is the main structural protein in cartilage. Scientific evidence has demonstrated the effectiveness of native (undenatured) type II collagen in supporting joint health and may therefore be used as an ingredient in consumer products.
However, the development of novel solutions with widely researched ingredients that are also easy to formulate is not a straightforward process. So how can manufacturers looking to make their next big move in the joint health market harness the powerful benefits of type II collagen?
Joint health: a growing market
The world’s population is ageing. Advances in healthcare and lifestyle improvements have contributed to increased longevity but as we age, our muscles, bones and joints can be impacted. In fact, 45% of individuals aged over 65 say they experience joint pain, which may affect their overall mobility and independence.[2] However, senior consumers are no longer taking mobility limitations sitting down. There is also an increasing interest in keeping fit and active as we age, with consumers starting to take a more proactive approach to protecting their joint health.
And it is not just the ageing population that are taking joint health more seriously. Several reports have shown that people who regularly play sport[3], the 40-plus population[4] and women experiencing menopause[5] also suffer joint discomfort or mobility issues. These trends have given significant momentum to the joint health sector and are a major driving force in the emergence of innovative joint health solutions. Between 2019 and 2024, it is forecast that the global bone and joint ingredients market will grow at a CAGR of 6.3% to meet this demand.[6]
Collagen for joint health
Glucosamine and chondroitin have long been used as active ingredients for joint health. Recently, collagen has been rapidly gaining market share as a result of rising consumer awareness of the health benefits it delivers. According to recent market data, sales in the joint health market increased by 4.3 % in 2018 in the US alone, largely driven by a boost in collagen sales, which increased by 30%.[7] As a result, the joint health category has experienced its highest overall growth since 2008.
Native type II collagen vs. hydrolysed type II collagen: what’s the difference?
Both native (undenatured) type II collagen and hydrolysed (denatured) collagen are used in commercial joint health products, but there are significant differences between the two forms.
In its natural form, collagen has a folded triple helix structure consisting of long polypeptide chains (see figure 1). Hydrolysed collagen is manufactured via a specific hydrolysis process, where enzymes “cut” the triple helix molecule into smaller pieces, i.e. short-chain peptides. This is why hydrolysed collagen is also known as collagen peptides, or denatured type II collagen. Native type II collagen is not hydrolysed and maintains its characteristic three-dimensional structure.
Figure 1. The triple helix structure of native (undenatured) type II collagen
Due to their structures, these two types of collagen also have different mechanisms of action. Native (undenatured) type II collagen works via an immune-mediated process, known as oral tolerance. Through this mode of action native type II collagen is recognised by the immune system as an endogenous substance, i.e. naturally occurring in the body, and deactivates the body’s immune response against its own collagen. On the other hand, hydrolysed collagen peptides are highly bioavailable, resulting in a source of the specific amino acids for de novo synthesis of collagen. As such, hydrolysed collagen peptides act as building blocks to maintain and rebuild cartilage.
Effectiveness at lower doses
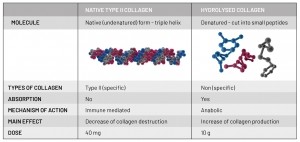
The daily dose and intake required for both collagens to be effective in the body varies greatly. The native (undenatured) type II collagen form is required at doses as low as 40 mg/day. Meanwhile, the recommendation for hydrolysed collagen is 10 g/day (see figure 2). The low dosage required for native type II collagen therefore mirrors consumer demand for easy-to-consume, convenient products - offering an innovative alternative to supplement manufacturers.
Figure 2. Native type II collagen vs. hydrolysed collagen
Exploring the link between immunity and joint health
Joint disorders involving inflammation and cartilage erosion, such as arthritic diseases, are characterised by an autoimmune component in which the immune system acts against the body’s own type II collagen.[8] This is because products from collagen breakdown are recognised by immune cells as potential pathogens and are therefore considered harmful to the body. As a consequence, an inflammatory and cartilage degradation response is activated. This involves an immune response against endogenous type II collagen, further damaging cartilage in the body.
Native type II collagen has been shown to help modulate the immune response against endogenous type II collagen through its mode of action, thus reducing joint inflammation and cartilage degradation.[9] When taken orally, native (undenatured) type II collagen passes through the stomach and reaches the intestine. It then interacts with the Peyer’s patches in the intestine, which are lymphoid nodules responsible for immune surveillance of the intestinal tract. This effectively turns off the immune response against endogenous type II collagen, helping to reduce joint inflammation and cartilage degradation.[10]
This mechanism of action means that only a small amount of native type II collagen to support joint health. This is why the standard dose of ingredients containing native collagen is just 40 mg, once daily, whereas dosages for hydrolysed collagen can be up to 10 g/day - an important benefit for manufacturers looking to meet the demand for effective, low-dose joint health solutions.
Mobilising the future with collagen
Clinical studies demonstrate the efficacy of b-2Cool, a native (undenatured) type II collagen developed by Bioiberica. This natural-origin ingredient is extracted from chicken sternum and is manufactured in controlled conditions to maintain its characteristic triple helix structure and the specific active epitopes of the native protein.
The properties of native type II collagen position it as an effective ingredient for joint health solutions that are convenient, low-dose and reduce pill fatigue. As manufacturers look to develop the next innovation in joint health supplements, native type II collagen therefore represents an exciting opportunity to make a measurable difference in the mobility market.
About Bioiberica:
Bioiberica is a global Life Science company specialised in the identification, extraction and development of biomolecules of high biological and therapeutic value for the pharmaceutical and nutraceutical industries. This specialisation has positioned Bioiberica as the leading Heparin API manufacturer and a world reference in the research, production and sale of other biologically-derived APIS and ingredients such as Chondroitin Sulfate, Glucosamine, Hyaluronic Acid, Native Type II Collagen or Thyroid. Since 1975, Bioiberica has consolidated its position as an expert in joint health and mobility thanks to a constant commitment to science and research.
These statements have not been evaluated by competent food authorities. This information is only for business-to-business use. The product is not intended to diagnose, treat, cure, or prevent any disease.
[1] M. Shoulders et al., ‘Collagen structure and stability’, Annu Rev Biochem., vol. 78, pg. 929-958, 2009.
[2] National Center for Health Statistics, United States, 2011, Hyattsville, MD, 2012.
[3] Cheng et al., ‘Journal of Clinical Epidemiology’, 2000, vol. 53, pg. 315-322.
[4] National Center for Health Statistics. Health, United States, 2011: Hyattsville, MD. 2012.
[5] Estudio MMYA, Rodriguez Bueno E. et al. Congreso AEEM 2010.
[6] https://mordorintelligence.com:81/industry-reports/bone-joint-health-ingredients-market
[7] NBJ Condition Specific Report 2019
[8] Kandahari, A. M. et al. Recognition of Immune Response for the Early Diagnosis and Treatment of Osteoarthritis. J. Immunol. Res., pg. 1–13, 2015.
[9] Park et al. Type II collagen oral tolerance; mechanism and role in collagen-induced arthritis and rheumatoid arthritis. Modern Rheumatology, vol. 19, no. 6, pg. 581-589, 2009.
[10] Ibid.