Promotional Features
NutraIso®: Meeting the Bioactive Dietary Needs for Nutritional Health and Wellness
While there are many plant-based nutritional extracts on the market, few provide the bioavailable absorption in the human digestive tract needed to assure nutritional efficacy.
Quintessence Nutraceuticals has successfully pioneered the development of a new all-natural plant derived nutraceutical nutritional for achieving bioavailability and bioactivity (efficacy) in meeting the holistic health and wellness needs of populations across a broad spectrum of demographics.
The Role of NutraceuticalsTrue nutraceuticals are natural nutritional supplements that are scientifically documented to provide health benefits beyond their basic aggregate nutritional values.[1]
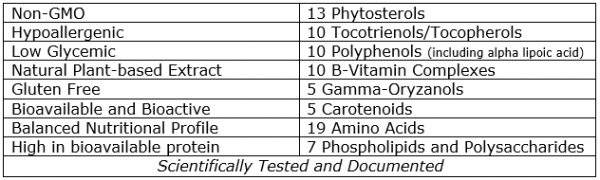
NutraIso® is a nutritionally dense extract derived from the bran and germ layers of brown rice. These hypoallergenic and gluten-free extracts are derived through the patented Quintessence enzymatic hydrolyzation process to yield a highly bioavailable and bioactive complex of phytonutrients with pharmacological values for mitigating many chronic health conditions which are often related to dietary imbalances in at-risk populations. For example, the NutraIso® proprietary process utilizes natural enzymes to hydrolyze complex proteins into simple proteins which are more digestible, thereby increasing protein bioavailability up to 2.5 times the typical aggregate protein content in the bran. Similar yield increases and bioavailability results are achieved from the enzymatic hydrolyzation of carbohydrates, dietary fiber and fat containing antioxidants embodied in the bran.
The Importance of BioavailabilityBioavailability by definition relates to the capacity of a nutritional isolate to be absorbed into the bloodstream, thus making the nutrient more bioactive (efficacious) in the human body for health and wellness.[2] To achieve bioavailability the aggregate complex nutrients must be broken-down into simple nutritional isolates for maximum absorption into the bloodstream. For example, complex proteins can be hydrolyzed into simple protein isolates comprised of peptides and amino acids for greater absorption into the bloodstream. Likewise, fats can be broken down into free fatty acids and antioxidant-rich oils that otherwise would be trapped in the fiber fraction and unavailable for the catalytic value they provide for enhancing bioavailability and bioactivity. Similarly, complex carbohydrates can be hydrolyzed into less complex polysaccharides for greater absorption. It is this enzymatic hydrolyzation breaking down of complex nutritional isolates that the patented NutraIso extraction method accomplishes.[3]
The proprietary Quintessence technology for enzymatic hydrolyzation provides a platform for extracting the highest level of bioavailable nutraceutical isolates from plant-based materials. Nutritional bioavailability and greater nutrient absorption into the blood stream are among the most sought-after product characteristics for nutritional functional foods in the current consumer-driven marketplace for enhanced health and wellness. NutraIso exceeds in providing scientifically documented bioavailable nutrition for meeting these consumer expectations.
NutraIso® Adult FormulationsThe aging population is increasingly confronted with a number of chronic health conditions that can be mitigated with scientifically documented nutraceutical dietary supplements that meet their personal nutritional needs. Scientific studies have concluded that chronic health conditions from prediabetic to early-stage cardiovascular health all have the potential to be managed through clinically validated dietary nutritional regimes.
For example, insulin dependence is typically the result of a progressive process of oxidative stress leading to insulin resistance and the eventual decline of pancreatic insulin production. This process can be mitigated with proper dietary intake and balanced nutrition. NutraIso® is the first scientifically documented nutraceutical dietary supplement to achieve patent status for reducing insulin resistance.[4],[5]
To this end, NutraIso® has been scientifically documented to lower fasting blood glucose and HbA1c levels in populations with early-stage health management concerns.
Recent clinical trials confirm that NutraIso effectively lowers Fasting Blood Glucose (mg/dl) an average of 23.1% and HbA1c levels to 5.97% in test subjects with average HbA1c levels of 7.03% at the beginning of the 90-day trial. Similar results were confirmed in test subject with HbA1c levels above 7.5% at the beginning of the trial. However, the most significant result of these trials was the increase in the insulin levels of among the test subjects. Those with HbA1c levels between 6.1-7.5% increased insulin levels an average of 40.37% during the 90-day trial, while those with HbA1c levels of 7.5% and above increased their insulin levels 123.49%. Moreover, among participants with virtually no insulin production in their initial fasting blood samples (0.92 uIU/ml), after 90 days of daily NutraIso supplementation insulin levels had increased to an average 13.32uIU/ml, well within the guidelines for healthy insulin levels. These results are statistically significant, scientifically documenting that bioactive nutraceutical supplements like NutraIso can play a critical role in mitigating the metabolic processes leading to insulin resistance. Insulin levels can actually be improved, thereby reducing insulin resistance in at-risk populations seeking to manage their dietary related chronic health conditions naturally. NutraIso® trials which focused on lowering cholesterol and triglyceride imbalances found similar results.
Driven by an ageing population, an increased incidence in adult chronic health conditions and a growing awareness for health and wellbeing, nutraceutical natural products are projected to grow at an estimated CAGR of 7.3%, reaching a market value close to $279 billion dollars by 2024.
NutraIso® Clinical Trial Results: Prediabetic and Type2 Diabetic
Infant NutritionWith chronic malnutrition in children reaching alarming levels globally, NutraIso® has the potential to play a paradigm-changing role in the health and wellbeing of infants worldwide.
It is currently estimated that malnutrition impacts the lives of nearly 165 million children globally under five years of age. Moreover, malnutrition is documented to have a crippling effect on the economic and socio-economic vitality of countries where malnutrition is most prevalent. These long-term impacts are largely the result of chronic malnourished infants at birth whose nutritional wellbeing was jeopardized during the mother’s pregnancy and lactation, thereby negatively and permanently impacting the child’s physical, cognitive and metabolic development. As a result, these children are sentenced to a lifetime of underperformance, poverty and food welfare dependence. The World Bank estimates that 3-5% of national GDP is lost to countries with high levels of infant malnutrition and, increasingly, developed countries are not immune from this debilitating chronic health condition.[6]
Quintessence Nutraceuticals has supported NutraIso® research and scientific documentation in infant nutrition through Sustainable Nutrition International, a 501(c)3 organization focusing on remediating chronic malnutrition in infants, particularly during the first 1000-day nutritional development window of opportunity.[7] NutraIso® formulations developed for lactating mothers during the exclusive breastfeeding period (EBF) have been tested and scientifically documented to significantly improve the health and wellness of EBF infants whose mothers consumed NutraIso® during lactation.[8]
Following World Health Organization protocols, NutraIso® formulations were found to significantly increase the physical and cognitive growth of EBF infants, showing a nutraceutical benefit translocation from mother to infant. These formulations will be further tested in 2019, but are currently available commercially.
NutraIso® Product CharacteristicsNutraIso® is an enzymatically hydrolyzed water-soluble nutritional supplement possessing nutraceutical isolates with pharmacological values for mitigating chronic health conditions.
CITED REFERENCES: [1] Medical Dictionary for Health Professionals and Nursing 2012.[2] Heaney, R.P. Journal of Medical Nutrition 2001.[3] Lynch, I.E., Sullivan, G.H. and Miller, L.R., U.S. Patent No. 8,945,642 B2 2015.[4] Reising, P.R. and Sullivan, G.H., U.S. Patent No. 9,192,180 B2 2015.[5] Sullivan, G.H. Sanchez, G.E. and Reising, P.R. (2015) How do NutraIso Nutritionals Work? White Paper.[6] Dr. Meera Shekar, Senior Nutritionist, World Bank Human Development Network.[7] Black, Robert E., et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. The lancet, 2013, vol. 382, no 9890, p. 427-451.[8] Sullivan, G.H. Sanchez, G.E. and Larry R. Miller (2002). Bioavailability and Bioactivity of NutraIso®. White Paper. SUPPLEMENTAL REFERENCES:1 Qureshi, A. (2002) Journal of Nutritional Biochemistry, 13: 175-187.2 Ryan, Elizabeth 2011, J. American Veterinary Medicine, 238 (5): 593-600.3 www.quintessencenutraceuticals.com4 https://sustainable-nutrition-intl.org/