Uterine fibroids are a widespread, significant health issue. Around 50% of women of reproductive age suffer from uterine fibroids, rising to even higher percentages in older people and women of color. Sufferers face symptoms including abnormal uterine bleeding and pelvic pain that negatively affect quality of life. Yet, the existing pharmacological and surgical treatment options have downsides that mean many women forego treatment. A new, better treatment option is needed.
The market opportunity and human need for a safe, effective option for the management of uterine fibroids is clear. Incidence of uterine fibroids, also known as leiomyoma, in reproductive age women ranges from 30% to 70%.1 As women age, incidence goes up. Uterine fibroids are even more common in some subpopulations. Incidence in women of color can be 80%, likely due to a mix of genetic and environmental factors.2
Around half of uterine fibroid cases are asymptomatic, although such people may have unrecognized symptoms including heavy uterine bleeding and fertility problems. In women who have symptoms, the condition manifests in a range of ways that are detrimental to their health and quality of life such as anemia and bladder pressure.
The severity of the symptoms leads some women to undergo hysterectomies to remove the fibroids. Development of additional surgical options including uterine artery embolization and radiofrequency thermal ablation enabled women to get relief from their fibroids without undergoing hysterectomies. However, those treatments still carry costs and complications, including potentially the persistence of the diminished fertility associated with uterine fibroids.3–5
Surveys show most women are concerned about the invasiveness and permanence of treatments for fibroids.6 The preservation of fertility is also important to many women. Those worries have spurred interest in pharmacological treatments for uterine fibroids that address the causes or symptoms of the benign gynecological tumors without requiring surgery and the costs and complications it entails.
The approval of ulipristal acetate, sold under the brand name Esmya, in the European Union in 2012 appeared to be a breakthrough in the availability of non-surgical treatment options for women with uterine fibroids.7 However, while the drug controlled bleeding in more than 90% of patients in clinical trials, evidence of severe side effects including liver injury emerged after the approval. European officials moved to revoke the approval in 2020.8 The US never approved the drug to treat fibroids.9
Validating a new, better treatment
The shortcomings of existing surgical and pharmacological treatments has created an opportunity for dietary supplements that can help manage uterine fibroids. Around two-thirds of women purchase over-the-counter products before seeking medical treatment for their fibroids, showing the demand for self-management options.6
Researchers responded to the unmet need by evaluating ingredients including vitamin D and green tea extract epigallocatechin gallate (EGCG) in women with uterine fibroids. The work generated some encouraging findings on endpoints including size reduction.
One clinical trial found vitamin D may stop, but not reverse, the growth of uterine fibroids.10 Another study linked vitamin D supplementation to a lower rate of progression to extensive disease.11 The trials built on evidence that low vitamin D levels are associated with more severe uterine fibroids.12 Separately, other researchers generated evidence that EGCG reduces uterine fibroid volume and the severity of symptoms.13
The body of research suggested vitamin D and EGCG may independently have some positive effects on uterine fibroids through mechanisms such as the downregulation of catechol-O-methyltransferase and inhibition of matrix metalloproteinase. However, there remained scope to improve on the effect either molecule achieved in isolation on key outcomes such as symptom improvement and volume reduction.
In 2020, researchers showed that combining vitamin D and EGCG may achieve better outcomes than either ingredient as a single agent.14 The study randomized 30 women with at least one fibroid more than 2 cm in volume to receive either no treatment or vitamin D, EGCG and vitamin B6 — in the form of Lo.Li. Pharma International’s Delphys dietary supplement — twice a day for four months.
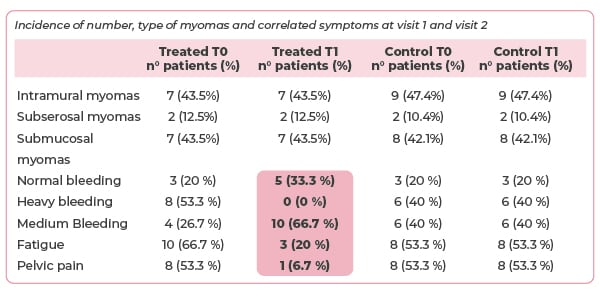
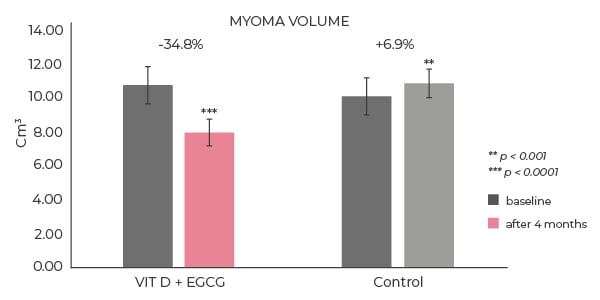
The total fibroid volume decreased by 34.7% in the treatment group, compared to a 6.9% increase in the control cohort (see graph 1). At baseline, 53% of women in the treatment group suffered from heavy bleeding (see table 1). None of the women suffered heavy bleeding after four months of treatment. Similarly steep declines in the incidence of fatigue and pelvic pain were seen over the course of the trial. In contrast, there were no reductions in the incidence of symptoms in the control group.
Addressing the unmet need
The clinical trial suggests Lo.Li.’s Delphys formulation of vitamin D and EGCG may address the need for treatments that bring symptom relief and reduce fibroid size without affecting fertility or causing other harms. Researchers are now building on those encouraging signs by gathering further evidence of the safety and efficacy of vitamin D and EGCG in the treatment of uterine fibroids.
Through the research, Italian pharmaceutical company Lo.Li. and its collaborators stand to provide a better treatment option for the many millions of women who suffer with uterine fibroids. In doing so, Lo.Li. will create a major new market opportunity for vitamin D and EGCG.
References
1. Vergara, D., Catherino, W. H., Trojano, G. & Tinelli, A. Vitamin D: Mechanism of Action and Biological Effects in Uterine Fibroids. Nutrients 13, (2021).
2. Wise, L. A. & Laughlin-Tommaso, S. K. Epidemiology of Uterine Fibroids: From Menarche to Menopause. Clin. Obstet. Gynecol. 59, 2–24 (2016).
3. Giuliani, E., As-Sanie, S. & Marsh, E. E. Epidemiology and management of uterine fibroids. Int. J. Gynaecol. Obstet. 149, 3–9 (2020).
4. Karlsen, K. et al. Fertility after uterine artery embolization of fibroids: a systematic review. Arch. Gynecol. Obstet. 297, 13–25 (2018).
5. Nam, J.-H. Pregnancy and symptomatic relief following ultrasound-guided transvaginal radiofrequency ablation in patients with adenomyosis. J. Obstet. Gynaecol. Res. 46, 124–132 (2020).
6. Stewart, E. A., Nicholson, W. K., Bradley, L. & Borah, B. J. The burden of uterine fibroids for African-American women: results of a national survey. J. Womens. Health 22, 807–816 (2013).
7. Tamayo, I. European Commission approves Esmya® for the pre-operative treatment of uterine fibroids (myomas). https://www.preglem.com/2012/02/27/european-commission-approves-esmya-pre-operative-treatment-uterine-fibroids-myomas/ (2012).
8. Czarska-Thorley, D. PRAC recommends revoking marketing authorisation of ulipristal acetate for uterine fibroids. https://www.ema.europa.eu/en/news/prac-recommends-revoking-marketing-authorisation-ulipristal-acetate-uterine-fibroids (2020).
9. Keown, A. FDA rejects allergan’s uterine fibroid treatment following EMA concerns over liver damage. https://www.biospace.com/article/fda-rejects-allergan-s-uterine-fibroid-treatment-following-ema-concerns-over-liver-damage (2018).
10. Arjeh, S., Darsareh, F., Asl, Z. A. & Azizi Kutenaei, M. Effect of oral consumption of vitamin D on uterine fibroids: A randomized clinical trial. Complement. Ther. Clin. Pract. 39, 101159 (2020).
11. Ciavattini, A. et al. Hypovitaminosis D and ‘small burden’ uterine fibroids: Opportunity for a vitamin D supplementation. Medicine 95, e5698 (2016).
12. Sabry, M. et al. Serum vitamin D3 level inversely correlates with uterine fibroid volume in different ethnic groups: a cross-sectional observational study. Int. J. Womens Health 5, 93 (2013).
13. Roshdy, E. et al. Treatment of symptomatic uterine fibroids with green tea extract: a pilot randomized controlled clinical study. Int. J. Womens Health 5, 477–486 (2013).
14. Porcaro, G., Santamaria, A., Giordano, D. & Angelozzi, P. Vitamin D plus epigallocatechin gallate: a novel promising approach for uterine myomas. Eur. Rev. Med. Pharmacol. Sci. 24, 3344–3351 (2020).


