Traditional ways of detecting iron deficiencies rely on blood samples – but this poses challenges in resource-limited settings away from labs.
The method uses an optical fibre probe on the inside of the lip to detect the faint fluorescence of red blood cell zinc protoporphyrin, a compound found in red blood cells when heme production is inhibited by lead or by a lack of iron.
The portable test takes around one minute to complete.
Dr Gary Brittenham, a researcher at Columbia University in the US and medical doctor at the New York-Presbyterian Morgan Stanley Children's Hospital, told us his team hoped the machines could be a pragmatic and hygienic way to tackle iron deficiency.
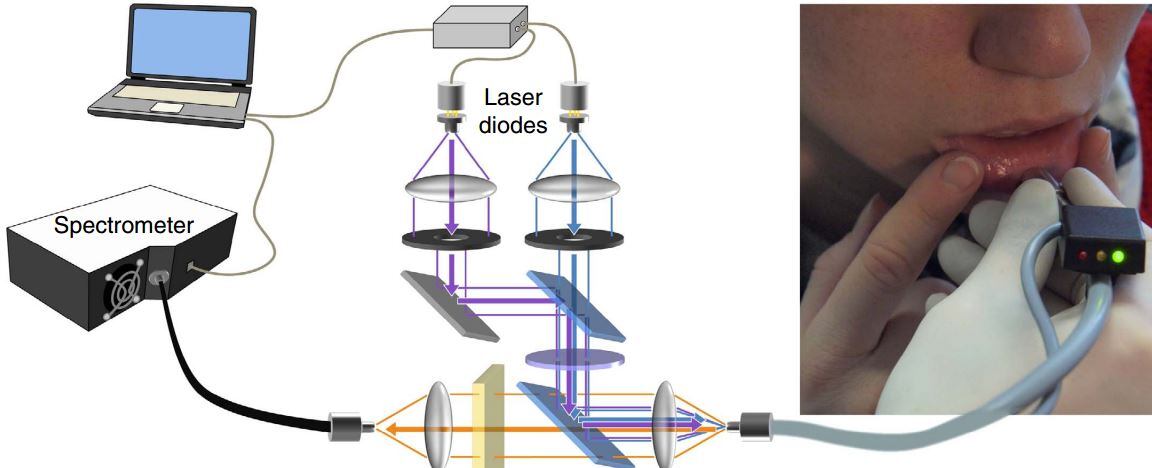
The results provide the first proof-of-concept for the non-invasive method, which was developed five years ago and has been supported for the past three years by almost a million euros from food giant Nestlé.
Published in Nature Communications, the initial study of 56 women suggested the method was comparable to the conventional haematofluorometer method, which traces zinc protoporphyrin in blood samples.
The researchers want to test the method in children and the general population.
Nadia Khaled, a project manager at Nestlé’s research arm Nestec and co-author of the paper, said Nestlé wanted to help “bring this wild idea to life” by bringing it up to this proof-of-concept stage.
Yet Dr Brittenham told us there was still a lot of work to be done before the machines could be sold to NGOs and national health services.
“We need to go further, we need to make this widely available.”
What’s next?
The group has secured a €600,000 grant from the German government to help develop and commercialise the idea as a start-up.
Worldwide iron deficiency affects more individuals than any other health problem.
"The numbers are staggering: 2 billion people – over 30% of the world’s population – are anaemic, many due to iron deficiency, and in resource-poor areas, this is frequently exacerbated by infectious diseases," according to the World Health Organisation (WHO).
In developing countries every second pregnant woman and about 40% of preschool children are estimated to be anaemic.
International patents for the technology are now pending and the researchers hope to get the current machine size of 30 x 20 x 15 cm down to the size of a paperback book.
It would likely take two years before the product could be commercialised, once all US and EU regulatory requirements to become a registered medical device were completed.
Once that happened, the anticipated selling price was about €8,000 for one machine.
Asked if this was a realistic cost for NGOs in developing countries, Dr Christian Homann, an expert in iron deficiency diagnosis at the University of Munich Hospital and co-author of the paper, told us it was a question of relative affordability.
“After the device is bought, only very minor costs arise, and for example no expensive calibration equipment is needed as for the hematofluorometer.”
Currently used hematofluorometers cost between $10,000 and $15,000 (€9,180 and €13,770) and require laboratory processing.
Other methods like high performance liquid chromatography can only be performed in specialised laboratories, which costs about €33 per single sample.
He said the €8,000 price of the new machines would largely account for material costs and margins were already “quite minor”.
“So we won’t be able to offer it significantly cheaper even in low-resource settings.”
However, if production was up-scaled to about 1000 devices per year, which they hoped would be possible within five years, the price could be reduced to between €3,000 and €5,000 per machine.
Diagnosing the solution
Dr Brittenham said his team’s project was inspired by a 2006 call from the World Health Organisation (WHO) for more detection methods for iron deficiencies in children, particularly in areas effected by malaria.
While doesn’t own any rights to this technology, Khaled said the project was in line with the company’s commitment to reducing iron deficiency, which it did largely through food fortification.
In 2014 Nestlé sold 183 billion servings of fortified foods worldwide, up from 167 billion in 2013. It has set itself the target of fortifying 200 billion servings of foods and beverages annually by 2016.
Head of Nestlé research communications Sarah Sheppard told us the project was part of an overall move towards diagnostics.
In September last year the company partnered with Swiss biotech AC Immune to develop a minimally invasive test for early-stage Alzheimer's disease.
This was part of a “catch and correct early” trend, Sheppard said.
“Definitely we’re looking at this more.”
Dutch nutrition giant DSM has also made moves in the area of alternative testing.
Last year a paper was published on its work with EU-backed project Food4me to use dried blood samples to test vitamin D statuses in 1465 participants from seven EU countries.
The samples were collected unsupervised in the home and the researchers highlighted the method as an efficient and accurate way to gather large sample sizes from around the world.
Source: Nature Communications
7:10776, DOI: 10.1038/ncomms10776
“Non-invasive detection of iron deficiency by fluorescence measurement of erythrocyte zinc protoporphyrin in the lip Georg Hennig”
Authors: C. Homann, I. Teksan, U. Hasbargen, S. Hasmuller, L. M. Holdt, N. Khaled, R. Sroka, T. Stauch, H. Stepp, M. Vogeser and G. M. Brittenham
