French firm Tetrahedron applied for novel food approval for its antioxidant marketed under the name Ergoneine back in 2013. Following a French food safety authority opinion, five other member states submitted concerns on the ingredient.
They asked whether adding the antioxidant to foods might be a contributing factor to rising rates of inflammatory diseases.
However, EFSA’s novel food opinion says the human studies submitted suggest there are “no relationships” between taking l-ergothioneine supplements and fortified foods and the susceptibility to or development of diabetes mellitus, Crohn's disease or rheumatoid arthritis.
EFSA’s Dietetic Products, Nutrition and Allergies (NDA) panel also said they had no concerns regarding genotoxicity at the proposed daily doses of 30 mg per day for adults and 20 mg per day for children in food supplements and foods like non-alcoholic drinks, cereal bars, milk, fresh dairy and chocolate.
Chemical reaction
As a natural compound, l-ergothioneine was first isolated from rye ergot (Claviceps purpurea) in 1909. It is naturally present in food sources like mushrooms, some varieties of black and red beans (Phaseolus vulgaris), offal and cereals.
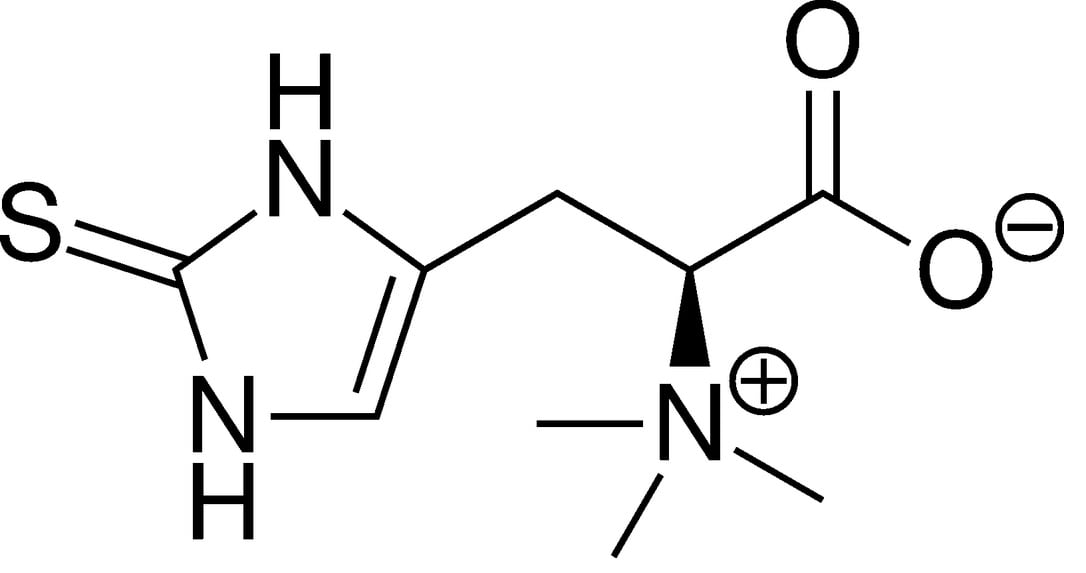
However Tetrahedron synthetically produces its l-ergothioneine through a patented process using water and a reaction between l-hercynine, bromine and cysteine. This industrial process allows the company to produce 250 kg of the ingredient each year.
President and co-founder of the Paris-HQ'd company, Jean-Claude Yadan, told us this chemical production, which mimics production of natural sources, held its own advantages.
“Because [l-ergothioneine] is of a chemical origin, the big advantage is that is pure. Those with biological origins – mushrooms for example – could contain other compounds more or less toxic such as heavy metals or toxins.
"It’s not better or worse, it’s different. But in terms of purity, it is advantageous to go chemical.”
He added that levels of l-ergothioneine in natural food sources like mushrooms could be significantly lowered by cooking.
Open for EU and US business
Yadan said the EU approval opened up exciting market opportunities for the novel synthetic antioxidant.
The company is now looking to secure B2B contracts in the EU.
In 2015 the company also secured US GRAS (Generally Recognised As Safe) status for the nutrient, which is the equivalent of the EU’s novel food approval.
Yadan said the company was preparing for transatlantic launch of supplements under the brand name Ergoneine at the beginning of next year.
These food supplements would initially contain only l-ergothioneine, but he said there was much scope to combine the antioxidant with other ingredients such as vitamins.
Asked what l-ergothioneine brought to the market over other antioxidants already available, he said: “In a few words, we can’t compare different antioxidants because each physiological antioxidant has a different function in the body.”

Targeting tissue
A review by researchers at the National University of Singapore’s Cardiovascular Research Institute published this year hypothesised that accumulation of ergothioneine is a way for the body to minimise oxidative damage.
Another paper by the same author, professor Barry Halliwell, also published this year suggested an age-related decline in blood levels of the antioxidant may be linked with cognitive health.
“Following absorption via a specific transporter, OCTN1, ergothioneine may accumulate preferentially in tissues predisposed to higher levels of oxidative stress and inflammation,” professor Halliwell and his colleagues wrote in the peer-reviewed journal Biochemical and Biophysical Research Communications.
“We found that whole blood ergothioneine levels in elderly individuals decline significantly beyond 60 years of age. Additionally, a subset of these subjects with mild cognitive impairment had significantly lower plasma ergothioneine levels compared with age-matched subjects. This decline suggests that deficiency in ergothioneine may be a risk factor, predisposing individuals to neurodegenerative diseases.”
