With the global GLP-1 market on track to hit $150bn by 2031, the clinical significance of glucagon-like peptide-1 is undeniable.1
This hormone’s multi-systemic influence on insulin secretion, gastric emptying and satiety signaling has become a major focus of metabolic research.
While GLP-1 receptor agonists have shown significant clinical efficacy, non-pharmacological options are being explored by researchers and ingredient developers.2 For industry innovators, the next frontier lies in research into endogenous GLP‑1 pathways: leveraging functional ingredients to stimulate the body’s natural hormonal response.
Understanding GLP-1’s role
GLP-1 is an incretin hormone produced primarily by the L-cells of the small intestine and released into the bloodstream after eating. Its downstream effects are wide-ranging: it promotes insulin release and reduces glucagon secretion from the pancreas, slows stomach release into small intestines to extend feelings of fullness and promotes digestive efficiency. Moreover, and most importantly for weight management, GLP-1 acts on the brain to influence appetite‑related signaling as part of normal physiological processes.³
This multi-organ reach is what has made GLP-1 such a compelling therapeutic target, and equally compelling as a focus for nutritional innovation. Certain bioactive compounds, including specific probiotic strains, polyphenol-rich plant extracts, and fermented botanical preparations have been investigated for their ability to influence endogenous GLP‑1 secretion under experimental conditions.3
The question for the nutrition industry has shifted from whether dietary GLP-1 modulation is possible to how such interactions can be characterised and measured in controlled research settings.

A multi-component approach to GLP-1 stimulation
TCI Biotech (TCI) has developed a proprietary GLP-1 Formula™ based on a synergistic combination of three primary actives: heat-killed postbiotic Bifidobacterium breve, kombucha black tea (Camellia sinensis) ferment, and Rockit apple extract. Each ingredient targets distinct yet interconnected signaling pathways along the gut-pancreas-peripheral tissue axis.
Bifidobacterium breve has been reported to stimulate the secretion of satiety hormones GLP-1 and Peptide YY (PYY) by activating G-protein receptors (GPR41/43), a response that persists even in heat-killed (postbiotic) preparations.4,5 These effects are supported by kombucha black tea ferment and Rockit apple polyphenols, which work to support insulin sensitivity and muscle glucose uptake by activating the AMPK and PI3K/Akt pathways while helping inhibit sugar-breaking enzymes.6,7
In one in vitro cell culture investigation, GLP-1 Formula™ demonstrated a 51.8% increase in GLP-1 secretion in human enteroendocrine cells, alongside a 79.8% upregulation of PYY gene expression.8
Moreover, changes in the expression of glucose transporter proteins (GLUT4 in skeletal muscle cells and GLUT2 in hepatocytes) were observed in vitro, which the authors described as indicative of coordinated cellular responses under laboratory conditions.⁸
Human evidence: Acute postprandial effects
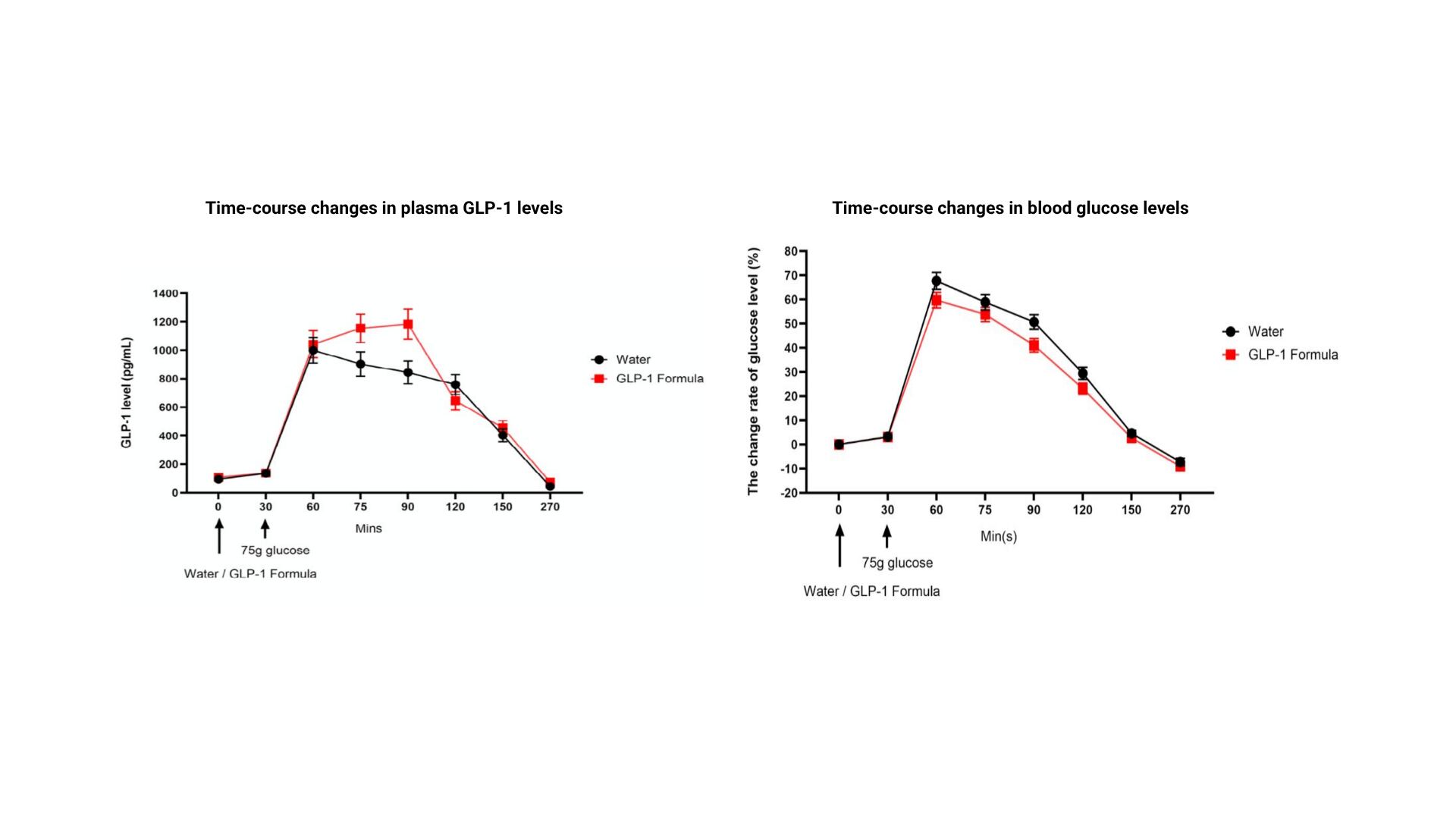
To translate these mechanistic findings, TCI conducted a randomised, controlled, crossover clinical trial in 25 adults with a body fat percentage greater than 25%.8 Participants consumed either 100mL of water or one bottle of GLP-1 Formula™ before a 75g oral glucose challenge, with a seven-day washout period between treatments. Plasma GLP-1, blood glucose, insulin, and subjective hunger and appetite scores were measured at multiple time points over a 270-minute postprandial window.8
Key observations from this study included: 8
- GLP-1 levels in the formula group were approximately 16% higher than controls before glucose ingestion, suggesting an early stimulatory effect of supplementation
- Post-glucose consumption, GLP-1 concentrations peaked at 75 to 90 minutes in the formula group, reaching increases of approximately 3.8-fold and 4.7-fold relative to the water control
- Unlike the transient GLP-1 surge observed with the control treatment, the GLP-1 Formula™ treatment presented a maintained elevated GLP-1 levels from 60 to 120 minutes, a sustained profile with potentially greater relevance for appetite suppression than a short-term peak
- Post-meal blood glucose excursions were attenuated at 60, 75 and 90 minutes, with relative reductions of approximately 8%, 5% and 9.7% respectively compared to the water control
- When participants received the GLP-1 Formula™, they reported lower subjective hunger and appetite (craving) scores throughout the postprandial period, with hunger scores approximately 23% lower than controls at the 150-minute time and trending toward better hunger control response
Real-world weight outcomes
Complementing the mechanistic data with a real-world practical application consumer study involving 560 participants evaluated weight outcomes following daily use of a GLP-1 Formula-based product during an eight-week period. Participants taking one serving daily before lunch reported to have lost an average of 4.9kg by the study endpoint, with over 59% losing at least 4kg.9
This consumer study published in Acta Scientific Medical Sciences in October 2024,suggested that a food-grade GLP-1-stimulating formula could support meaningful weight management.9
This formulation offers favourable tolerability profile, specifically addressing the gastrointestinal sensitivities that often limit the use of intensive pharmacological interventions. While the open-label, unblinded format of the study lends to a follow up randomised, double-blinded, placebo control trial format. TCI has two studies underway in 2026 in Taiwan and the US.
A growing category with growing evidence
The convergence of consumer awareness around GLP-1, the widespread adoption of metabolic health tracking technologies and increasing demand for sustainable weight-management solutions has created significant market momentum for food-grade GLP-1 modulators. Market analysts suggest that the ‘GLP-1 companion’ and ‘booster’ categories are likely to see increased consumer interest as metabolic health tracking becomes more mainstream.10
As the metabolic health category matures, the focus is shifting to research‑driven, physiology‑aligned investigations.. While pharmaceutical GLP-1 agonists remain a primary clinical tool for obesity, there is an evident market for non-pharmacological options that support the body’s endogenous hormonal pathways.
The current data on multi-component, food-grade formulations, such as the combination of heat-killed probiotics and botanical polyphenols suggest areas for continued investigation into postprandial GLP‑1 dynamics under defined study conditions.
While large-scale, long-term longitudinal data remains the gold standard for clinical validation, the existing human trials and real-world observations provide a promising foundation. As further results from upcoming studies emerge, the industry will gain a clearer understanding of how these nutritional strategies can best serve as a complementary approach to long-term weight management and metabolic wellness.
References
- The Economist. New drugs could spell an end to the world’s obesity epidemic.
- Holst, JJ. The incretin system in healthy humans: the role of GIP and GLP-1. Metabolism. 2019;96:46-55.
- Yue Y.; et al. Effect of food ingredients on glucagon-like peptide-1 secretion in STC-1 and HuTu-80 cells. Int J Food Sci Technol. 2019;54(12):3214-3222.
- Greiner, TU.; et al. Microbial regulation of GLP-1 and L-cell biology. Mol Metab. 2016;5(9):753-8.
- Sugahara, H. Differences between live and heat-killed bifidobacteria in the regulation of immune function and the intestinal environment. Benef Microbes. 2017;8(3):463-472.
- Fang, W. Theaflavins in black tea ameliorate high-fat diet-induced obesity and inflammation via gut microbiota, AMPK-mediated metabolism, and NF-kappaB pathway. Phytomedicine. 2025;148:157314.
- Manzano, M. Apple polyphenol extract improves insulin sensitivity in vitro and in vivo in animal models of insulin resistance. Nutr Metab (Lond). 2016;13:32.
- Huang, SC.; et al. Modulation of Postprandial GLP-1 Secretion and Glycemic Responses by a Food-Grade GLP-1 Formula in Human Subjects. J Obes Weight Loss Ther. 2026;16:878.
- Chi-Fu, C.; et al. Efficacy of GLP-1 Formula on Body Weight in Overweight and Obese Adults. Acta Scientific Medical Sciences. 2024;8(10):62-67.
- Innova Market Insights. Top Ten Food and Beverage Trends 2026.



