Liposomal formulations are becoming increasingly popular in the nutraceutical industry thanks to their ability to boost absorption of essential nutrients.
The global liposomal supplement market size is expected to grow from $377.39m in 2025 to $745.02m in 2034.1 Its growth is attributed to rising consumer demand for high-bioavailability supplements, particularly those supported by clinical evidence and regulatory validation.
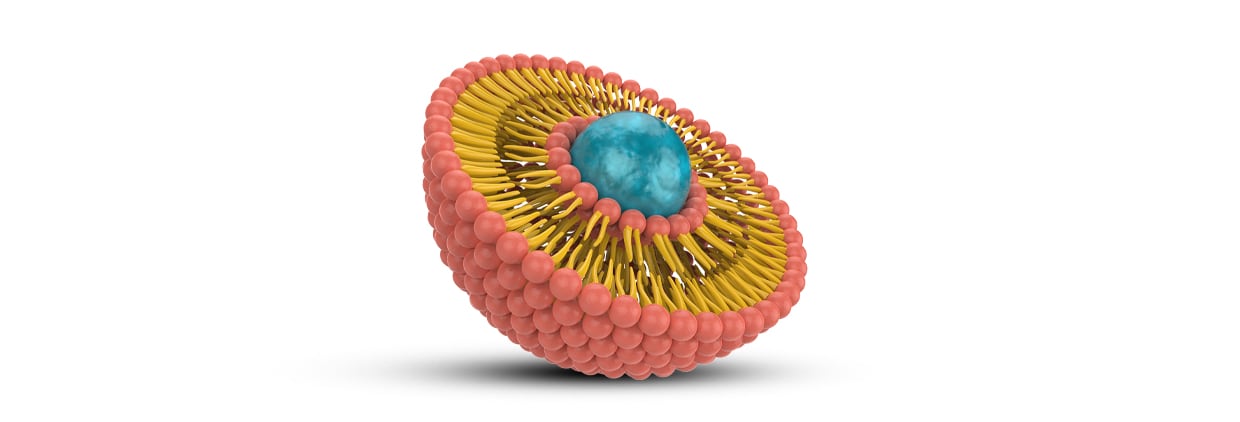
As a spherical artificial membrane with phospholipid bilayers, a liposome has a membrane-like property which closely resembles the structure and functionality of natural biological cell membranes, allowing it to serve as a highly biocompatible and versatile vehicle for drug delivery.
As these vesicles are similar to the cell membranes found in the human body, they provide a Trojan Horse effect that shields nutrients from degradation, improves absorption and stability and enhances bioavailability.
Most of the nutraceutical supplements face a common challenge – limited bioavailability. No matter how potent the ingredient is, if it doesn’t reach the cells effectively, the impact will be minimal. This is where liposomal systems come in.
By mimicking the human cell membranes and protecting the compounds from harsh environments, liposomal systems are particularly relevant for poorly absorbed nutrients, including vitamin C and Coenzyme Q10 (CoQ10) and molecules prone to degradation or crystallinity.
Current market challenges
While liposomal systems can effectively increase bioavailability, practical applications can be limited. Common issues of liposomal products consist of poor stability, vesicle collapse during drying, low encapsulation efficiency, rapid release and degradation in the gut. In fact, some products show ‘liposomal-like claims’ without the structural integrity, clinical evidence or demonstrated bioavailability improvement that is required.
So, what makes a ‘true’ liposomal product? Developing a liposomal product is not just about technology; it’s about precision and purity. A ‘true’ liposomal product can be defined by the presence of microscopic, spherical phospholipid bilayer vesicles that encapsulate nutrients. Authentic nano-sized liposomes mimic cell membranes to enhance absorption. This requires sophisticated manufacturing techniques, rather than simple blending.
Clinically supported liposome technology
Molecules Biolabs is India’s premier liposomal research and innovation hub. It develops cutting-edge nutraceuticals driven by a multi-disciplinary team of scientists and industry leaders in chemistry, molecular biology and pharmaceutical sciences.
Blending traditional wisdom with modern science, Molecules Biolabs works to address the complex challenges in the health and nutrition industry. In doing so, Molecules Biolabs has created Metazome technology, a next-gen liposomal solution tailored to address the major limitations of conventional liposomes.
Metazome™ is an established liposomal platform, developed through continuous refinement and application across multiple formulations. Built on sustained research and iterative optimization, it reflects a technology that has evolved with advancing scientific understanding to deliver consistent performance.
Metazome technology encapsulates nutrients within microscopic lipid layers to enhance absorption and protect delicate compounds from degradation, enhancing bioavailability. From sourcing premium raw materials to employing rigorous manufacturing processes under stringent quality controls, every step of the production line is designed to meet the highest global standards.
Vitamin C: Supporting applications in nutraceuticals and functional foods
Vitamin C, also known as ascorbic acid, is a water-soluble micronutrient. Supplementation of vitamin C is important as humans cannot synthesize this essential nutrient, making dietary intake crucial for collagen production and immune function.
Vitamin C has potent antioxidant properties with essential roles in immunity, collagen synthesis, nutrient absorption and enzymatic cofactor activity in hydroxylation reactions. It can help to prevent deficiency diseases like scurvy, aids in iron absorption and supports skin, blood vessel and bone health.
Using Molecules Biolabs Metazome technology, a study published in 2025 analysed the development of a stabilised liposomal vitamin C formulation which organises gum arabic nanospheres within powdered liposomes.2
Results highlighted that Metazome vitamin C (MVC) showed nanosized spherical morphology, high encapsulation efficiency and strong negative zeta potential. MVC also exhibited sustained release (≈58% at 4 h, pH 2.0; ≈51% at 4 h, pH 6.8) and retained >90% vitamin C after 180 days at 4 and 25°C.
Additionally, a randomised, double-blind crossover clinical trial showed that MVC achieved a 7.62-fold higher bioavailability, 6.29-fold higher Cmax, and longer t1/2 than conventional vitamin C, supporting applications in nutraceuticals and functional foods.
CoQ10: Improving oral bioavailability
CoQ10 is a vital nutrient naturally produced by the body. Found in every cell, particularly the heart, liver and kidneys, it creates energy (ATP) and acts as a powerful antioxidant. As levels decline with age, supplementation is important to help support heart health, reduce fatigue and promote healthy ageing.
CoQ10’s oral efficacy can be limited by low aqueous solubility, high crystallinity and poor bioavailability. Although several lipid-based and nanoformulations have been explored, many suffer from limited stability, incomplete suppression of crystallinity or modest pharmacokinetic improvement.
A study published in 2026 study aimed to develop a stable, scalable liposomal CoQ10 formulation and evaluate its physicochemical properties and human oral bioavailability.3 Metozomal CoQ10 (MCQ) was developed using Metazome technology, in which CoQ10 was incorporated into phospholipid bilayers reinforced with gum arabic nanospheres and converted into a dry, reconstitutable liposomal powder by spray drying.
Results showed that MCQ significantly improved pharmacokinetics compared with conventional CoQ10 (CCQ), including a 4.3-fold increase in AUC0−t, a 3.6-fold increase in Cmax, prolonged Tmax, extended half-life, and a lower elimination rate constant (p < 0.01).
The study highlighted that integrated Metazome-based architecture represents a key innovation by combining amorphization, nanoscale liposomal delivery and structural stabilisation. This resulted in superior stability and markedly enhanced human bioavailability.
Importance of patented and clinically-proven liposomal ingredients
Patented and clinically-proven liposomal ingredients are vital to the nutraceutical industry, acting as a differentiator for consumers. Patented, clinically-proven ingredients can guarantee bioavailability, while providing documented safety and stability standards – distinguishing them from unverified, low-quality alternatives.
Molecules Biolabs’ Metazome is a patented, stable, dry liposome technology tailored to address the limitations of conventional liposomes. It uses an advanced stable liposomal formulation for superior bioavailability, helping to transform the nutraceutical industry with an ingredient clinically-proven for superior absorption.
References
- Precedence Research. Liposomal Supplements Market Size and Forecast 2025 to 2034.
- Amalraj, A.; et al. Development of Liposomal Vitamin C Stabilized by Gum Arabic Nanospheres Using Metazome Technology: Characterization, In Vitro Release, and Oral Bioavailability of Vitamin C in a Randomized, Double-Blind, Single-Dose, Two-Period, Two-Way Crossover Study. ACS Nutrition Science. 2025; 1 (1).
- Amalraj, A.; et al. Next-generation liposomal coenzyme Q10: from formulation to clinical evidence via metazome technology for improved stability and enhanced oral absorption. Food and Function. 2026; Issue 3.