Akkermansia muciniphila has emerged as one of the most promising next-generation probiotics due to its well-documented role in gut barrier integrity, metabolic health, and immune modulation.
Originally isolated from the human gut, this anaerobic bacterium is unique in its ability to utilize mucin as a primary energy source, making it highly specialized and consequently, challenging to cultivate and stabilize.
Despite its growing interest in the nutraceutical and functional food space, the commercialization of live Akkermansia muciniphila presents several technical hurdles. Its strict anaerobic nature, sensitivity to oxygen, and dependence on mucin-like substrates make both large-scale production and stability in finished formats particularly difficult. These challenges have driven innovation in fermentation strategies, culture media, and, most critically, stabilization technologies such as microencapsulation.
What is microencapsulation?
Microencapsulation technologies can be broadly categorized into physical, physicochemical, and chemical methods, including spray drying, freeze-drying, extrusion, emulsification, and coacervation. Among these, spray drying is the most widely used technique in the probiotic industry due to its scalability and cost-effectiveness.
The selection of encapsulating materials plays a critical role in determining the protective efficiency and release behavior of the system. Common wall materials include polysaccharides (e.g., alginate, starch, inulin), proteins (e.g., whey protein, gelatin), and lipid-based carriers. These materials are chosen based on their ability to form stable matrices, provide barriers against oxygen and moisture, and support microbial viability during processing and storage.
Microencapsulation systems can also be engineered for controlled or targeted release. In probiotic applications, this involves designing matrices that remain stable under the acidic conditions of the stomach while enabling the gradual release of viable cells in the intestine, where pH and enzymatic conditions are more favorable for survival and colonization.
Recent advancements in microencapsulation have focused on improving encapsulation efficiency, minimizing cell leakage, and enhancing resistance to bile salts and digestive enzymes. These improvements are particularly important for next-generation probiotics, where maintaining functional viability is essential for achieving desired health outcomes.
Microencapsulation refers to the process of enclosing active ingredients, such as probiotic bacteria, within a protective coating or matrix at the microscopic level. This encapsulating layer serves as a physical barrier against environmental stressors, including oxygen, moisture, temperature fluctuations, and mechanical stress.
In probiotic applications, microencapsulation provides protection during manufacturing processes (e.g., drying, blending, and compression), enhances stability during storage and transportation, and improves survivability under harsh conditions, including oxygen exposure, as well as during gastrointestinal transit.
For oxygen-sensitive organisms such as Akkermansia muciniphila, microencapsulation is essential to preserve viability throughout production, storage, and delivery, ensuring functional activity upon administration.
Measuring probiotic potency
Probiotic potency is traditionally measured in colony forming units (CFU), which quantify the number of viable microorganisms capable of forming colonies under defined growth conditions. While CFU remains the industry standard, it presents limitations when applied to next-generation probiotics like Akkermansia muciniphila, due to its strict anaerobic growth requirements, slow growth kinetics, dependence on mucin or mucin-like substrates, and if it’s microencapsulated.
Akkermansia muciniphila is notoriously difficult to quantify using conventional plating methods. Even minor deviations in environmental conditions, particularly oxygen exposure, can significantly impact viability and lead to underestimation of true potency.
Akkermansia muciniphila enumeration: A specialized approach
To address these challenges, specialized enumeration methods are required. Vidya, a leading manufacturer of herbal extracts and natural wellness supplements, has developed a proprietary standard operating procedure tailored specifically for Akkermansia muciniphila. The process involves several critical steps:
- De-encapsulation: The protective matrix surrounding the bacteria is carefully disrupted to release viable cells without causing damage.
- Pre-activation: The bacteria are rehydrated and activated in peptone water to restore metabolic activity.
- Selective culturing: The activated culture is introduced into a nutrient-rich medium consisting of Brain Heart Infusion (BHI) supplemented with mucin or mucin-like substrates to support growth.
- Anaerobic incubation: Samples are incubated at 37°C under strictly controlled anaerobic conditions for approximately 24 to 72 hours.
- Colony enumeration: Resulting colonies are counted to determine viable CFU levels.
This targeted methodology ensures accurate and reproducible quantification while overcoming the limitations of standard probiotic testing methods.
Enhancing stability through microencapsulation
One of the most significant barriers to commercializing live Akkermansia muciniphila is maintaining stability throughout processing and shelf life. Exposure to oxygen, even for short durations, can drastically reduce viability. Additionally, moisture and temperature fluctuations further compromise product integrity.
Microencapsulation directly addresses these issues by creating an oxygen barrier that protects cells during manufacturing and storage, reducing moisture ingress (which can trigger degradation), improving thermal stability during processes such as spray drying while also enhancing survivability in complex formulations and delivery formats.
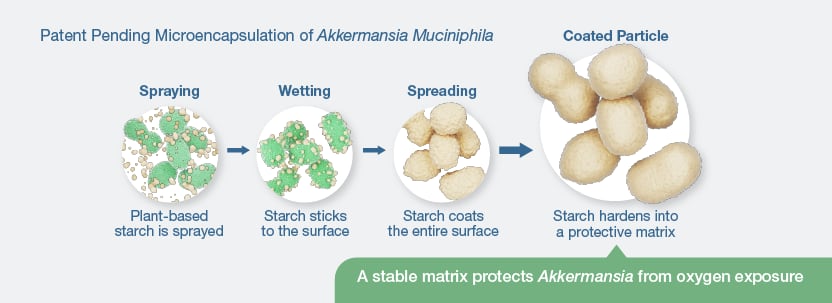
Vidya’s approach incorporates encapsulation using tapioca starch, forming a protective matrix around the bacterial cells that not only shields the organism from environmental stress, but also enables the production of a stable, free-flowing powder that will be suitable for large-scale applications.
Advancing Akkermansia muciniphila manufacturing with novel fermentation and encapsulation
Beyond encapsulation, advancements in upstream fermentation processes play a crucial role in achieving both high yield and stability. A key innovation lies in the replacement of traditional animal-derived mucin with plant-based alternatives. Since Akkermansia muciniphila relies on mucin as a nutrient source, developing a structurally similar plant-based substrate is essential for scalable production.
Vidya has developed a plant-based gum system that closely mimics the structure and functionality of mucin. This innovation eliminates the need for animal-derived inputs, reduces cost and ethical concerns, and supports robust bacterial growth under controlled conditions. Using this approach, fermentation is conducted under anaerobic, high-cell-density conditions with tightly controlled parameters such as pH, temperature, agitation, and CO₂ levels.
This optimized process significantly improves efficiency, reducing fermentation time from approximately 72 to 80 hours to 36 to 40 hours while achieving high potency levels (≥100 billion CFU per gram). Following fermentation, the biomass is harvested via centrifugation, encapsulated, and spray-dried to produce a stable final product.
Advanced manufacturing and stabilization technologies
As interest in next-generation probiotics continues to grow, so does the need for advanced manufacturing and stabilization technologies. Akkermansia muciniphila represents both an exciting opportunity and a significant technical challenge due to its unique biological requirements.
Microencapsulation has emerged as a key enabling technology, providing the protection necessary to maintain viability throughout production, storage, and delivery. When combined with innovations in fermentation such as plant-based mucin analogs and high-efficiency processing these advancements are making the large-scale commercialization of live Akkermansia muciniphila increasingly feasible.
Together, these technologies are paving the way for more stable, effective, and accessible probiotic products, ultimately supporting the next wave of innovation in gut health and beyond.





