S‑equol is one of the few bioactive molecules that meaningfully connects three key themes in women’s health: premenstrual Syndrome (PMS) in younger women, menopausal transition in midlife, and long‑term healthy aging.
It is neither a traditional hormone nor a generic plant extract. Instead, it is a gut‑derived metabolite with highly selective receptor‑modulating activity.
This article explores these three dimensions to systematically illustrate how S-equol, through its role as a signal modulator provides targeted support for women at every stage of life.
The overlooked daily burden: Connection between S‑equol and PMS
For many women, the days before menstruation come with a familiar set of discomforts – irritability, mood dips, headaches, sleep disturbances, cravings, and difficulty focusing. These symptoms fall under the medical definition of PMS, while more severe cases are classified as Premenstrual Dysphoric Disorder (PMDD).1
The prevalence is striking. Up to 90% of women experience at least one premenstrual symptom. Around 20% to 30% meet the criteria for PMS, and 2% to 5% meet the diagnostic threshold for PMDD.2 With rising stress, irregular routines, and dietary shifts, PMS is becoming increasingly common and increasingly disruptive.
Clinical evidence linking equol status to PMS severity
Existing PMS treatments – such as oral contraceptives or Selective Serotonin Reuptake Inhibitors (SSRIs) – can be effective but often cause side effects, motivating researchers to explore gentler regulatory pathways.2 This is where S‑equol stands out.
Multiple studies have demonstrated a meaningful association between S‑equol and PMS:
- 2016 cross‑sectional study: Among women aged 20 to 45, 41.8% of healthy participants were equol‑producers, compared with only 23.9% among women with PMS.3 This suggests that women capable of naturally producing S-equol have significantly lower PMS risk.
- 2018 study in female athletes: Among Japanese female athletes aged 18 to 21, 54.5% experienced performance impairment due to PMS symptoms. Non‑equol producers showed a substantially higher risk (OR = 3.34), indicating that S-equol may play a regulatory role even in highly active individuals.4
- 2022 open‑label intervention: Nineteen non‑equol‑producing women with PMS supplemented 5mg S‑equol daily, starting on day 5 of their cycle for two consecutive cycles. Symptoms significantly improved: 10.5% achieved a 50% reduction in Daily Record of Severity of Problems (DRSP) score, with notable improvements in depression, anxiety, irritability, and mood swings.5
- 2026 Japanese clinical study: Among healthy women aged 19 to 20, the proportion of equol‑producers was significantly lower in those with moderate‑to‑severe PMS/PMDD.6

Together, these findings reveal a clear pattern: for women who cannot produce S-equol naturally, S‑equol supplementation offers a safe and effective pathway to relieve PMS by modulating receptor signaling rather than altering hormone production.
The midlife turning point: How S‑equol reshapes menopausal health management
If PMS represents cyclical discomfort during the reproductive years, menopause is a systemic transition driven by sustained estrogen decline. It affects not only vasomotor symptoms like hot flashes but also skin integrity, bone density, cardiovascular function, and neurological health.
Declining estrogen disrupts several biological networks:
- Skin: Reduced fibroblast activity and down-regulated collagen‑related genes (e.g., COL1A1), leading to loss of elasticity and hydration.7
- Neurological: Reduced GABA and serotonin regulatory capacity disturbs sleep, with 40% to 56% of postmenopausal women reporting persistent sleep issues.8
- Metabolic and vascular: Altered lipid metabolism, decreased arterial elasticity, and increased chronic inflammation risk.9
These interconnected pathways collectively accelerate aging.
Selective ERβ activation: The smart key mechanism of S‑equol
The unique value of S‑equol lies in its receptor selectivity. The body has two primary estrogen receptors:
- ERα: Found mainly in breast and uterine tissue; overstimulation may raise proliferative risk.
- ERβ: Located in the brain, bones, vascular endothelium, and skin; activation supports vasomotor stability, bone health, cardiovascular resilience, and neurological protection.
S‑equol has approximately nine‑fold higher affinity for ERβ than ERα, unlike estradiol used in hormone therapy, which activates both receptors similarly.10
This makes S‑equol a smart key, preferentially unlocking beneficial pathways while minimizing stimulation of sensitive tissues.
Clinical evidence: Benefits from hot flashes to bone health
S‑equol has accumulated robust human data:
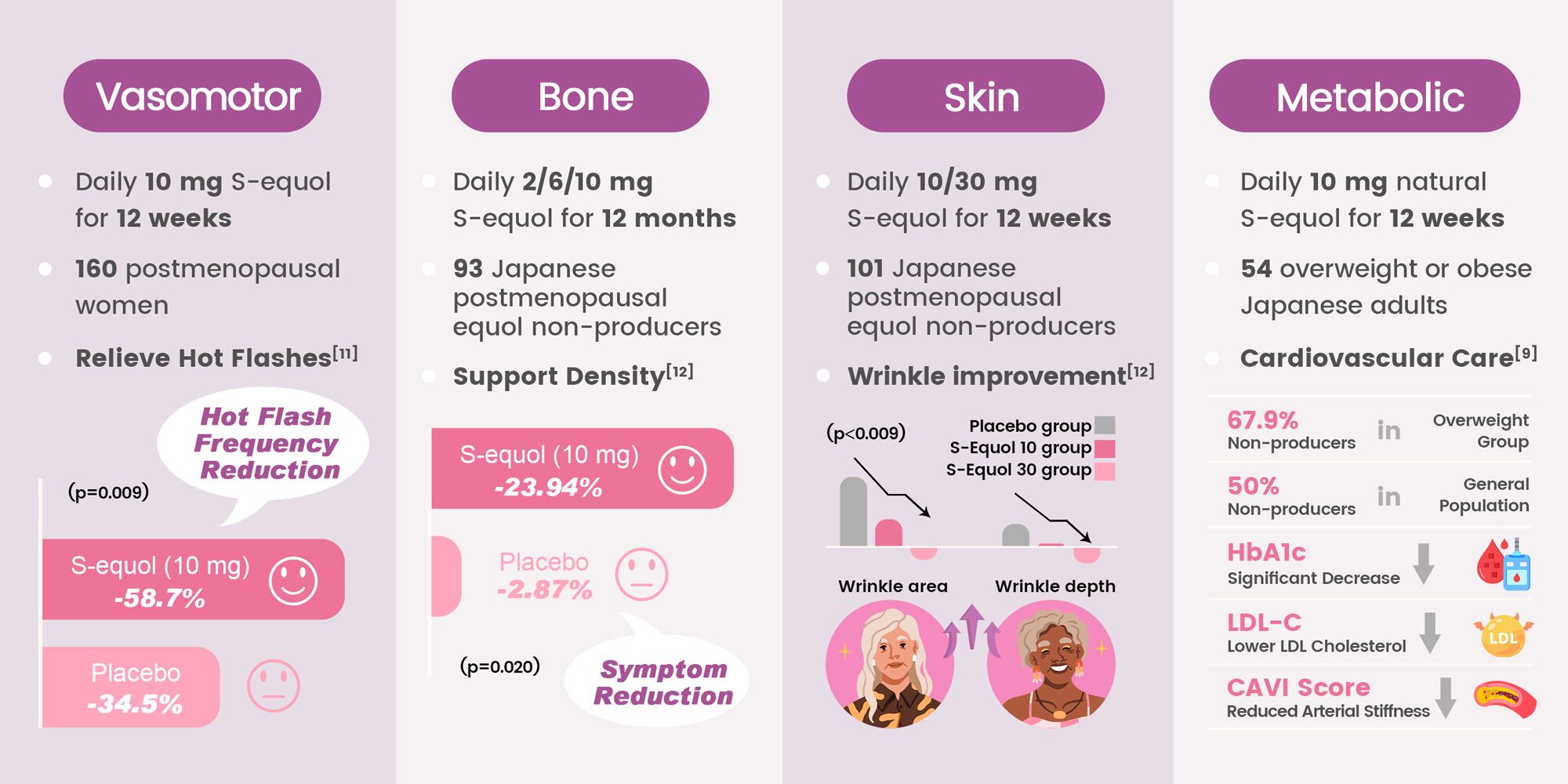
- Vasomotor symptom relief: A randomized, double‑blind study in 161 menopausal women found that 10mg S‑equol daily for 12 weeks significantly improved hot flashes and neck‑shoulder stiffness. Among women with ≥3 daily hot flashes, improvements were 39.2% greater than placebo.11
- Bone health support: In postmenopausal non‑equol producers, 10mg daily for 12 months reduced bone resorption markers and helped slow bone density loss.12
- Skin improvement: Supplementation with 10mg or 30mg for 12 weeks reduced wrinkle area; the 30mg dose also reduced wrinkle depth, without affecting hormone levels or gynecological parameters.13
Cardiovascular and metabolic effects S‑equol improved arterial stiffness (CAVI) and enhanced lipid profiles by lowering the LDL‑C/HDL‑C ratio.9 These findings show that S‑equol supports sleep, cardiovascular health, bone maintenance, metabolic balance, and skin quality, making it a comprehensive tool in menopausal care.
A new anti‑aging paradigm: From hormone replacement to signal modulation
Soy isoflavones have long been used as natural estrogen‑like compounds, but their true activity relies on being converted into S‑equol by gut bacteria. Only 30% to 50% of Asians and 20% to 30% of Western populations have this capability.14
This variability explains inconsistent consumer experiences and limits the effectiveness of soy isoflavones as a reliable intervention.
Breakthroughs in fermentation technology
Natural plant sources contain only trace amounts of equol precursors, and extraction can be inefficient. Industrialization became feasible only with advances in microbial fermentation.
Companies such as Bonerge Lifescience have developed specialized strains and patented technology capable of producing high‑purity, human‑identical S‑equol at scale. This ensures high bioactivity, consistent supply, and full control over stereochemistry – something chemical synthesis cannot guarantee.
Fermentation breakthroughs opened the door to commercializing S‑equol as a functional ingredient.
From raw material to complete solution
Bonerge’s EquoYouth™ S‑equol offers a high‑credibility ingredient supported by:
- Pure S‑configuration: Fermentation‑derived S‑equol with >99% purity, identical to the body’s endogenous form.
- Safety certification: Recognized by the US FDA with Self-Affirmed GRAS status.
- Clinical commitment: Supported by human studies to build a solid efficacy‑and‑safety evidence base.
- Reliable supply: Large‑scale production with stringent quality controls.
Brands choosing EquoYouth gain not only an ingredient but a full scientific and operational framework.

A lifelong anchor for women’s health
S‑equol is expanding beyond its traditional association with menopausal support:
- For reproductive‑age women, it offers safe relief for PMS and mood fluctuations.
- For perimenopausal women, it helps reduce hot flashes and preserve bone and cardiovascular health.
- For women focused on long‑term healthy aging, it represents a shift from hormone replacement to precise signal modulation.
From adolescence to postmenopause, S‑equol is emerging as a core functional molecule that bridges hormonal transitions and systemic aging.
In a field driven by evidence, meaningful long‑term solutions must demonstrate clear mechanisms, strong data, and defined applications. S‑equol meets these criteria – positioning ingredients like EquoYouth at the forefront of next‑generation women’s health innovations.
References
- Henshaw C A. PMS: diagnosis, aetiology, assessment and management. Revisiting… Premenstrual syndrome. Advances in Psychiatric Treatment. 2007; 13(2): 139-46.
- Management of Premenstrual Disorders: ACOG Clinical Practice Guideline No. 7. Obstetrics and gynecology. 2023; 142(6): 1516-33.
- Takeda, T.; et al. Relation between premenstrual syndrome and equol-production status. The journal of obstetrics and gynaecology research. 2016, 42(11): 1575-80.
- Takeda, T.; et al. Premenstrual symptoms interference and equol production status in Japanese collegiate athletes: A cross-sectional study. The journal of obstetrics and gynaecology research. 2018; 44(3): 488-94.
- Takeda, T.; et al. Evaluation of a natural S-equol supplement in treating premenstrual symptoms and the effect of the gut microbiota: An open-label pilot study. Neuropsychopharmacology reports. 2022; 42(2): 127-34.
- Kada-Kondo, N.; et al. Urinary Equol Production Capacity, Dietary Habits, and Premenstrual Symptom Severity in Healthy Young Japanese Women. Metabolites. 2026; 16(1).
- Vijayakumar, V.; et al. S-equol status modulates skin response to soy isoflavones in postmenopausal women: results from a randomized placebo-controlled pilot trial. Frontiers in nutrition. 2025; 12: 1671835.
- Fidecicchi, T.; et al. Neuroendocrine mechanisms of mood disorders during menopause transition: A narrative review and future perspectives. Maturitas. 2024; 188: 108087.
- Usui, T.; et al. Effects of natural S-equol supplements on overweight or obesity and metabolic syndrome in the Japanese, based on sex and equol status. Clinical endocrinology. 2013; 78(3): 365-72.
- Kuiper, G.G.; et al. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta. Endocrinology. 1997; 138(3): 863-70.
- Aso, T.; et al. A natural S-equol supplement alleviates hot flushes and other menopausal symptoms in equol nonproducing postmenopausal Japanese women. Journal of women’s health (2002). 2012; 21(1): 92-100.
- Tousen, Y.; et al. Natural S-equol decreases bone resorption in postmenopausal, non-equol-producing Japanese women: a pilot randomized, placebo-controlled trial. Menopause (New York, NY). 2011; 18(5): 563-74.
- Oyama, A.; et al. The effects of natural S-equol supplementation on skin aging in postmenopausal women: a pilot randomized placebo-controlled trial. Menopause (New York, NY). 2012; 19(2): 202-10.
- Setchell, K.D.; et al. S-equol, a potent ligand for estrogen receptor beta, is the exclusive enantiomeric form of the soy isoflavone metabolite produced by human intestinal bacterial flora. The American journal of clinical nutrition. 2005; 81(5): 1072-9.