Responding to EFSA’s April, 2012, rejection of its article 13.5 submission under the EU nutrition and health claims regulation (NHCR), Merck said bioavailability and efficacy data should be reconsidered, as EFSA’s health claims panel had misinterpreted it the first time around.
But EFSA’s Dietetic Products, Nutrition and Allergies (NDA) panel dismissed the Merck appeal as ungrounded and reiterated its earlier opinion “that a cause and effect relationship has not been established between the consumption of glucosamine and maintenance of normal joint cartilage.”
The NDA rejected arguments raised by Merck relating to healthy versus diseased study populations; bioavailability; and biomarkers and demonstration of physical effects.
Merck was not available for comment about the significance of the NDA verdict at the time of publication.
Grounds for rejection
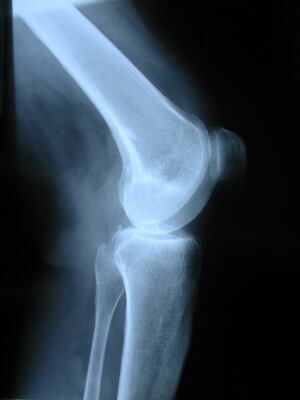
Of bioavailability concerns, the NDA stated that, “while the available evidence shows that some dietary glucosamine is taken up into blood and synovial fluid, the Panel considered that uptake of glucosamine into cartilage cells would be very limited under in vivoconditions.”
It said in vitro and animal studies showing joint health mechanisms was “weak”, and not necessarily able to, “predict an effect of glucosamine on joint cartilage in humans.”
Urinary concentrations of type II collagen fragments were not valid biomarkers to predict joint cartilage degradation.
On study group extrapolation it affirmed, “no conclusions could be drawn from the studies on patients with osteoarthritis for the scientific substantiation of the claim in subjects without osteoarthritis.”
In February 2011, the UK Advertising Standards Authority (ASA), censored a marketing campaign for Merck Consumer Healthcare-owned Seven Seas JointCare range for overstating benefits.
Merck Consumer Healthcare markets a number of food supplements under different brands including Seven Seas based in the UK, and Merck Médication Familiale based in France.
This Merck rejection follows a 2009 article 13.1 general function opinion that glucosamine does not benefit the normal function of joints, either alone or in conjunction with typical food supplement stable mate, chondroitin.
Seven Seas products are sold in the UK, Europe, south east Asia and latin America.
