Set to be debuted at the CPhI 2013 industry event in Frankfurt this month, the product will be available initially in Italy - where Chr. Hansen said that around half of probiotic units sold come in this liquid shot format - before taking the product global in 2014.
“We are really looking forward to showcasing this healthy, lemon-flavoured shot that combines two ingredients which both have a long history of use, high consumer awareness and acceptance,” said Chr. Hansen’s Lasse Nagell, sales and marketing vice president, health and nutrition.
The 10ml shots will be a combination of Chr. Hansen’s “flagship” probiotic strain BB-12 (Bifidobacterium) and fiber in the form of fructo-oligosaccharides (FOS).
Liquid opportunity
Nagell told NutraIngredients that Chr. Hansen has previously produced liquid probiotics in an oil drop form but that this was targeted more specifically at the infants and young children nutrition segment. “This will be our first liquid format going out broader as a supplement,” he said.

Yet opportunities within this child-friendly market, as well as with elderly consumers, remain key to this launch. ““Instead of taking a pill or a capsule, a small liquid shot is of course easier to administer,” Nagell explained. “We know that using Italy as a reference this is a very popular dosage form for children.”
A liquid form also presents greater opportunities in terms of taste profiles, he said, which would be vital in ensuring acceptance from children. “It’s very easy to work with flavours with liquid, maybe easier when you look at adding flavour and profiling to tablets.”
For this initial Italian launch the probiotic will be available in lemon flavour only, but Nagell said the company’s manufacturing partner Biofarma has the capabilities to work with any flavour.
Drycap technology
The drinkable probiotic shot will come in a mini bottle using Drycap technology which allows live probiotics in a dry environment.
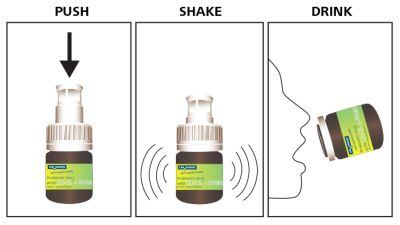
Nagell said the firm had overcome issues of stability through this alu-blister packaging technology integrated into the mini-bottle cap and can offer probiotics with a shelf life of 24 months.
Health claims
The BB-12+ fiber mix can be promoted as a 2 in 1 or dual action product which Chr. Hansen says addresses gastrointestinal function and irregularities.
The European Food Safety Authority’s rulings on probiotic health claims have been the source of some contention in recent years, prompting the food science agency to issue guidance to companies wishing to submit a probiotic health claim application last month.
“Outside of Europe I think there are tremendous opportunities to work with local health claims,” Nagell said.
