Consumers for Health Choice (CHC), an opponent of unified EU MPLs it views as restrictive and damaging to the UK and other liberal markets that allow high-dose products, circulated the Brussels-based Food Supplements Europe (FSE) memo supporting MPL imposition at the first opportunity.
CHC director of strategy Chris Whitehouse told us lobbying efforts were intensifying ahead of European Parliament elections on May 22-25 next year, and the possible replacement of Tonio Borg as European Commissioner for Health and Consumer Affairs (DG SANCO) later in the year.
Under Borg, the EC has shown no inclination to advance the issue in the face of strong lobbying from CHC and other groups.
“What they want will seriously damage the UK market and that certainly wouldn’t be in the interests of consumers in those markets that are more liberal,” Whitehouse said.
“They want to get in with new MEPs and force a rushed and unfortunate decision, or to force Borg to take some action.”

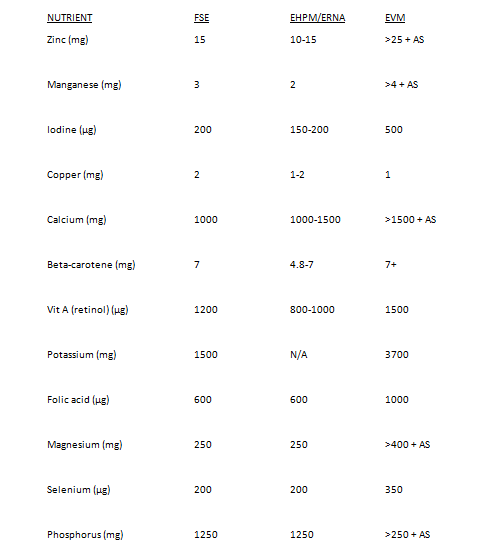
The leaked documents included FSE’s MPL recommendations Whitehouse described as, “pretty horrendous from the UK point of view.” Those recommendations included vitamin B6 at 18mg per day and calcium at 1000mg per day.
FSE, formed earlier this year, counts large manufacturers like Merck, Bayer, Amway and Herbalife, along with nutrient suppliers like BASF and DSM among a membership that also includes smaller players.
The leaked document, titled ‘Priority Dossiers involving the European Parliament’, states EU MPL harmonisation should proceed in harmony with 2002 EU Food Supplements Directive (FSD) safety criteria and established risk assessment and intake knowledge.
“The legal deadline has passed and it is not likely that a decision will be taken by the current Commissioner for Health Tonio Borg.”
“This means that today different levels are applicable between the Member States with no apparent justification, hindering free movement of products and choice of consumers.”
“FSE is seeking support from the European Parliament to urge the Commission to proceed with harmonisation and the setting of MPL based on these safety criteria and Member States to support this approach.”
Other groups like the Alliance for Natural Health International (ANH-I) are critical of this safety model and has joined with Dutch research house, TNO, to develop a new model that will be ready by the summer of 2014.

ANH-I: “…low doses suit them fine…”
ANH-I executive and scientific director, Robert Verkerk, PhD, said there was “nothing new” in the FSE push for harmonisation.
“The big companies selling vitamins and minerals into pharmacy and supermarkets across the EU have always wanted full harmonisation of maximum levels because it makes life much easier for them,” Dr Verkerk said.
“They’ve also never been greatly interested in the benefits of their products so low doses suit them fine. For many of these companies, their core interest is pharmaceuticals.”
He added: “The approaches that have so far been considered by the Commission are over 10 years old and omit some of the latest scientific developments. We’re expecting to see outputs from TNO towards the middle of next year and once these fresh data are on the table, that’ll be the best time to really engage governments and elected representatives.”
FSE was not available for comment at the time of publication.