Naturalpha has launched a white paper on the subject as well as a clinical trial platform that could connect companies with research resources such as hospitals in its home town of Lille.
Discussing why it had chosen this as its first topic-specific platform, the company said NAFLD was a growing medical issue with an estimated worldwide prevalence of 20% in the general population and 40-90% in obese people.
Despite this growing prevalence, neutraceutical players were yet to step up to this challenge en masse.
Eric Chappuis, the firm’s director of consulting, said: “Our feeling is that this topic, which is poorly addressed to date by the nutrition and nutraceutical industry, will become a major topic in the coming years. However the lack of understanding of the epidemiological burden of these conditions as well as sometimes short-term vision of the nutra industry has not yet led to concrete development.”
For this to be unlocked though, commitment to clinical trials was needed.
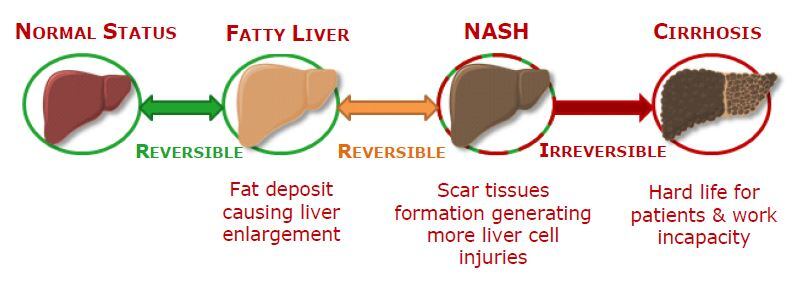
Non-alcoholic fatty liver diseases (NAFLD) is a term used to describe liver diseases involving hepatic steatosis – or fatty change – without excessive alcohol intake and includes a wide range of liver diseases from simple fatty change to non-alcoholic steatohepatitis (NASH). NASH causes liver cirrhosis – scarring of the liver and subsequent loss of function due to long-term damage – and may develop into hepatocellular carcinoma, the most common type of liver cancer.
Gathering momentum
Vitamin E could be one area of interest, Chappuis said, as well as probiotics.
This month meta-analysis of five studies on the impact of vitamin E on NAFLD and non-alcoholic steatohepatisis (NASH) was published in the journal Nutrition.
The Japanese researchers said the results showed vitamin E could significantly improve liver function in patients with fatty liver diseases including NASH. The research caught the eye of Dutch supplement firm DSM, which said it would help inform clinical guidelines for practitioners.
Meanwhile, Nestlé published an international patent this month on pomegranate skin extract for fatty liver disease, for which it cited research with rats.
What can you say?
Given the nature of the condition, Chappuis said companies would of course have to be wary of making disease claims.
He envisaged nutrition supplements marketed in a similar way to plant sterols and stanols in cholesterol-lowering spreads – i.e. a nutritional product meant to compliment medical strategies.
A preventative positioning could also be possible.
Currently there was only one liver health claim approved by the European Food Safety Authority (EFSA) stating: “Choline contributes to the maintenance of normal liver function”.
Since the implementation of the 2006 nutrition and health claims regulation (NHCR) marketing experts have noticed firms reversing the direction of NPD – looking at what claims can be made legally in the EU then working backwards to the product idea.
Chappuis acknowledged this was the easiest route to take but while the risks were higher with NAFLD so were the potential gains.
