Crescendo Organics and Natural Products is seeking ‘substantial equivalence’ – EU parlance for a product virtually the same as another approved ingredient or product – for its Argentine-grown chia with already approved chia grown in Australia by The Chia Seed Company.
The UK Food Standards Agency (FSA) Advisory Committee on Novel Foods and Processes (ACNFP) is calling for input on the application until November 3 this year.
Crescendo wants to use its chia seeds in bread products, breakfast cereal, fruit, nut and seed mixes and seed packs.
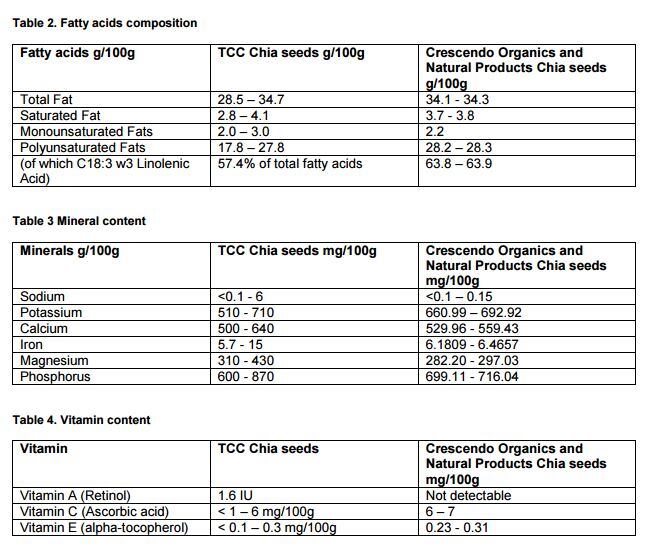
“We have noticed some slight differences in nutrient content (protein, magnesium and vitamin C) but these do not seem significant in our view,” it said in its application.
It noted a maximum daily intake of 15 g.
Chia, here
Chia seeds require novel foods approval because they were not consumed to a significant degree before 15 May 1997. The first approval was granted to Robert Craig & Sons in 2009.
Many others have followed for the ‘supergrain’ including those from Australia, Canada, Chile and the UK.
‘Substantial equivalence’ means similarity in composition, nutritional value, metabolism, intended use and level of undesirable substances to an approved product across the EU’s 28 member states.
Novel foods are expected to be reformed and streamlined and to encompass the likes of nano materials in a vote in the European Parliament next week. If passed, a 2-year transition period will follow.
Chia seeds are a source of protein, fibre, vitamin B forms riboflavin, niacin, thiamine and minerals such as calcium, phosphorous, potassium, zinc and copper.