The acquisition, expected close in four months, is set to “double Probi from a financial perspective”, Probi’s chief executive officer (CEO) Peter Nählstedt said in an online financial hearing this morning from Sweden.
Probi chief financial officer (CFO) Niklas Brandt said the buy was a “very exciting opportunity” that would increase Probi’s in-house manufacturing capacity, improve its strain delivery technology and put the company in a strong position for future acquisitions in the probiotic area.
Washington State-based Nutraceutix holds a patent for delivery technology BIO-tract which protects probiotics from stomach acids and meters intestinal release as well as the manufacturing process LiveBac which enables the manufacturing of tablets with ‘sensitive ingredients’ like probiotic strains, according to the company.
Brandt said this would help it increase the efficiency of its products which contain its strains selected for pH sensitivity.
Improved survival rate in the stomach thanks to Nutraceutix’s technology could help Probi charge a premium for its products, use more sensitive strains and in the future even reduce effective clinical doses, he said.
Fastest growing probiotics market
The company said the trans-Atlantic purchase was strategic, with North America representing one of the fastest growing probiotic markets in the world.
According to Euromonitor International figures, in 2014 North America accounted for 47% of the global probiotic food supplements market while Europe accounted for 31%.
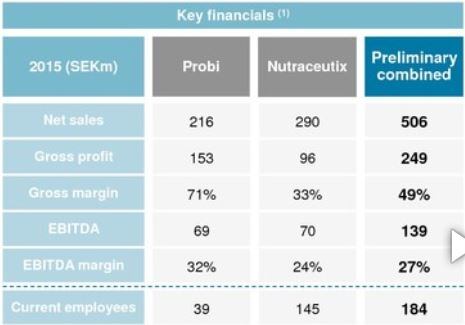
With 173 additional US customers promised through the buy, Probi predicts its North America net sales will triple.
“To ensure growth for a probiotics company we believe the right strategy is to have a strong exposure in North America,” Brandt said.
Marketing of probiotics in the EU is notoriously difficult, with the term itself outlawed as an unauthorised implied health claim under the EU nutrition and health claims regulation (NHCR).
Probi is one of the latest companies to apply to the the European Food Safety Authority (EFSA) for a specific health claim. It expects a decision on its claim that “Lactobacillus plantarum 299v increases non-heme iron absorption” by July this year.
Yet 300+ probiotic health claim submissions have so far been rejected, dating back to 2008 when the Parma, Italy-based agency issued the first opinions under the newly written NHCR.
Until such a claim is approved the only legal option to EU marketeers is to simply reference the Latin strain name on pack with perhaps a mention of the number of colony forming units (CFU) and leave consumers to do their own homework.
350m tablets, 4.3m bottles

Probi currently relies solely on third-party manufacturing.
Brandt said the acquisition would increase its in-house manufacturing capacity, which had clear financial benefits, but there was still scope to increase its third-party orders to keep up with growth plans.
Nutraceutix produces about 350 million tablets and 4.3 million bottles of products every year and made SEK 290m (€31.43m) in net sales in 2015.
This marked a “small decline” in sales for the company following the retraction of one customer’s product resulting in a loss of $7m (€6.16m), which it was only able to partially recover through other clients.
Integration philosophy
The joint company will be headquartered in Lund in Sweden with manufacturing facilities remaining in the US and R&D and global marketing in Sweden for the Probi- and Nutraceutix- branded products.
Brandt said job security and an “integration philosophy” was key to the success of the new venture, so that “people were not focused on losing their jobs” but on becoming a leading probiotics business.
The combined staff head count will be 180 people.
In the deal, Probi was advised by Handelsbanken Capital Markets, Mannheimer Swartling Advokatbyrå AB, Hjalmarsson & Partners Corporate Finance AB and Wiggin and Dana LLP. Nutraceutix was advised by Cascadia Capital and Perkins Coie LLP.
