The supplements were tested against the limits in the GOED Voluntary Monograph, which are stricter than most pharmacopeial standards and global regulations. When compared to the less stringent limits applied by the European and British Pharmacopeias and the Australian authorities, the rates of compliance would be even higher. The data is published in Scientific Reports.
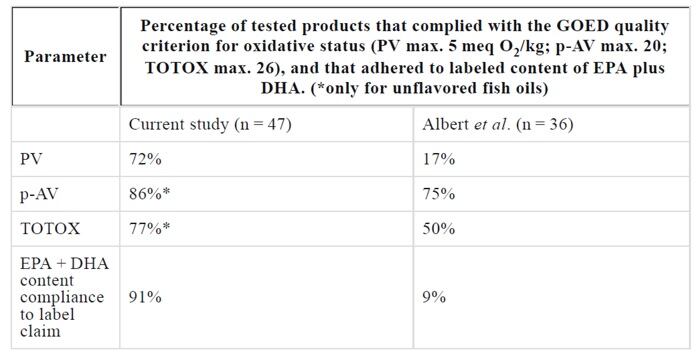
The results are in stark contrast to findings published in January 2015 by Albert et al (Scientific Reports, 5:7928), which claimed that only 9% of the fish oils in the New Zealand market met label claims for EPA (eicosapentaenoic acid) and DHA (docosahexaenoic acid), and 17% were below PV limits (a measure of oxidation).
“The current study provides a high level of confidence in the accuracy of the obtained test results since multiple accredited laboratories tested the same samples, and obtained acceptable reproducibility in the results on oxidative status and omega-3 LCPUFA content,” wrote the authors, led by Gerard Bannenberg, Director of Compliance and Scientific Outreach for the Global Organization of EPA and DHA Omega-3s (GOED).
The study was performed by representatives from GOED, DSM Nutritional Products, Omega Protein, Eurofins Central Analytical Laboratories, Nutrasource Diagnostics Inc, and Alkemist Labs.
“These results indicate that most, but clearly not all, of the analyzed fish oil products comply with standards for oxidative quality and EPA/DHA content voluntarily set by GOED. The current findings concur with results from third-party product quality monitors, such as Consumer Reports, Labdoor.com, ConsumerLab.com, and the International Fish Oil Standards Program, all of which report their test results publicly and note that a majority of fish oil supplements are compliant with oxidation limits and EPA/DHA label claims,” added the authors.
Replication of results
The Albert results garnered a lot of media attention at the time, particularly in New Zealand, but the new GOED study is the fourth (and the largest) that was unable to replicate the original Albert results.
Specifically, the Therapeutic Goods Administration (TGA) in Australia tested 15 products in their local market and found 100% compliance with Australian regulatory limits, The Omega-3 Centre conducted its own tests on a sample of 10 products from Australia and similarly found 100% compliance with regulatory limits, and more recently, a newly-developed laser testing method was used to test 11 New Zealand products and found that all met label claims.
Study details

For the current study, GOED purchased every fish oil supplement available from online pharmacies in New Zealand and sent blinded samples to multiple independent labs for testing. The samples were tested against the self-imposed limits in the GOED Voluntary Monograph.
The results showed that 91% of the products complied with EPA/DHA content claims.
In addition, 72% of the products complied with GOED maximum limits for primary oxidation, as measured by the Peroxide Value (PV of less than 5 meq O /kg). Eight-six percent complied with limits for secondary oxidation, as measured by para Anisidine Value (pAV of less than 20), added the authors, while 77% complied with TOTOX limits, which is a combined measurement of total oxidation (the GOED limit is less than 26). The data is presented in the table below.
“If the less stringent limits for pAV, PV and TOTOX used by the European and British Pharmacopeias and the Australian authorities would be held as reference in the current study, even higher rates of compliance can be concluded. For example, 100% of the tested fish oils complied with a pAV limit of 30, 98% with a PV limit of 10 meq O /kg, and 96% with a combined TOTOX value of 50,” explained the authors.
“In other words, the reported level of product compliance in published studies depends on the regulatory framework used, but no evidence for a largescale noncompliance of fish oil supplements in New Zealand is found with regard to oxidation or EPA/DHA content.”
“Consumer can feel confident in the quality of the fish oil products”
GOED analysis
For a comprehensive analysis by Adam Ismail, GOED's executive director, please click HERE.
Commenting on the discrepancy with the results of Albert et al. the authors noted that, informatively, the data from the earlier study showed high conformity with secondary oxidation levels, but not primary oxidation levels. This would suggest recent oxidation in the samples, perhaps linked to taking too long to prepare the samples for analysis or unsuitable handling in the lab.
Another reason could be the testing of flavored oils because flavorings are known to skew oxidation results, added the researchers.
GOED’s Bannenberg said: “This replication study results shows that fish oil supplements on the New Zealand market contain the correct content of EPA and DHA, and are not excessively oxidized. Consumers can feel confident that fish oils they can purchase are quality products.”
Bannenberg noted that nearly 100% of fish oil products in New Zealand complied with Australian and New Zealand regulatory limits for oxidation and EPA/DHA content claims and that compliance was similarly high with stricter, self-imposed limits set by the international industry.
To learn more about the GOED monograph, its origins and its impact on quality in the industry, please click HERE.
Source: Scientific Reports
2017, 7: 1488, doi: 10.1038/s41598-017-01470-4
“Omega-3 Long-Chain Polyunsaturated Fatty Acid Content and Oxidation State of Fish Oil Supplements in New Zealand”
Authors: G. Bannenberg, C. Mallon, H. Edwards, D. Yeadon, K. Yan, H. Johnson, A. Ismail

