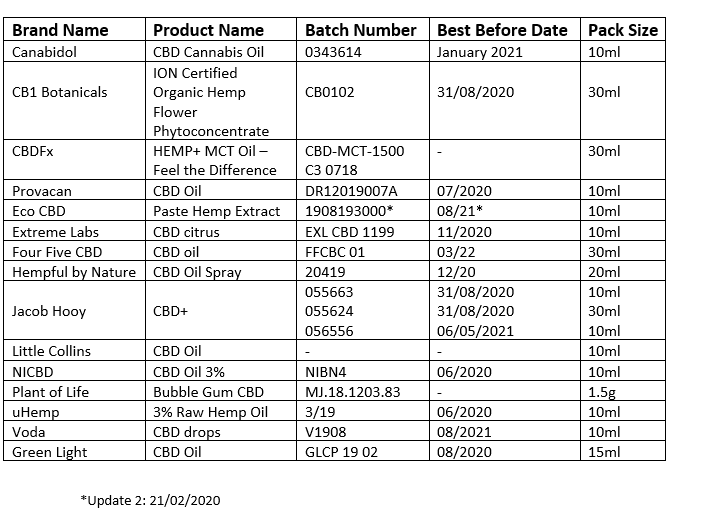
The updates now add one additional product to the recall product list and the batch number (1908193000) and 'best-before' date (08/21) for another product on the list, Eco CBD Paste Hemp Extract.
“The implicated batches of CBD foods and food supplements contain unsafe levels of delta‐9‐tetrahydrocannabinol (THC) based on the European Food Safety Authority (EFSA) acute reference dose,” the recall update sates.
“There are no immediate dangers associated with the consumption of these products.”
For food businesses, including online retailers currently selling the implicated batches of the CBD products, the FSAI orders them to be immediately removed from sale.
The authority also ask the retailer to contact their supervising environmental health officer (or foodincidents@fsai.ie) so that any remaining stock can be removed.
“Food businesses that have sold the implicated batches of the below CBD products to consumers must display a point-of-sale notice in store and on websites if sold online, to inform consumers that the implicated batches of these CBD products are being recalled and the reason why,” the authority adds.
Consumer advice
Additional advice by the FSAI also advised consumers not purchase or consume any of the implicated batches of the below CBD foods and food supplements.
Environmental health officers (EHOs) were also requested to check food businesses to determine if implicated batches of the below CBD products are available for sale.
“If found, EHOs should seize, remove and detain the products off site; also oversee a recall, ensuring a point-of-sale notice is displayed in store and on websites if sold online,” the notice states.
The recall comes after the FSAI carried out a survey earlier this month that revealed 37% of the products tested had a tetrahydrocannabinol (THC) content that could exceed safety limits set by the European Food Safety Authority (EFSA).
In addition, over 40% of samples varied significantly (over 50%) from declared CBD content, which the FSAI says, places consumers at risk of “being grossly misled to being put at risk by the ingestion of relatively high levels of THC”.
It also found 34% (13/38) of the samples were classified as novel foods and thus required authorisation before being placed on the EU market.
Further findings revealed that 36% (13/36) of samples classed as food supplements had not been notified to the FSAI before being placed on the market, as required by the law.
Many of those notified also have issues to be addressed, such as notifying changes of labels.
Untenable situation
Retailers in the CBD space naturally expressed frustration at the survey, dubbing the situation “untenable”.
“The discrepancy, with all of this, relates to where on the spectrum the TRACE amounts of THC sit and where that sits within Food Law, and other relevant law,” says Ireland-based retailer Little Collins CBD Dispensary.
“There exists CLEAR and SUBSTANTIAL confusion between current FSAI testing methods, accepted practice and indeed prior communications from said Government body.
“The situation remains untenable; it is grossly unjust to punish the end user and the retailer for a lack of clear framework and governance in the emerging cannabis industry in Ireland.
“We are disappointed at the lack of prior consultation from the FSAI. In the cannabis industry, trust is everything and such a broad-brush statement from a State body, effectively tarnishing every company on the list they published, is extremely unwarranted in the circumstances.”
The FSAI’s CEO Dr Pamela Byrne said the survey was carried out to determine exactly how compliant these products were with EU and Irish food legislation.
She added that people were consuming CBD products where quality control was poor with some possibly not getting what they are paying for, unwittingly exposing themselves to psychoactive THC.
“Also, of concern is that people consuming CBD products containing significantly high levels of THC could fail a drug test and the implicated batches of the products identified in the survey are now subject to a product recall,” she added.
“We are working with the Environmental Health Service of the HSE in relation to other products identified in the survey and further appropriate action will be taken.”




