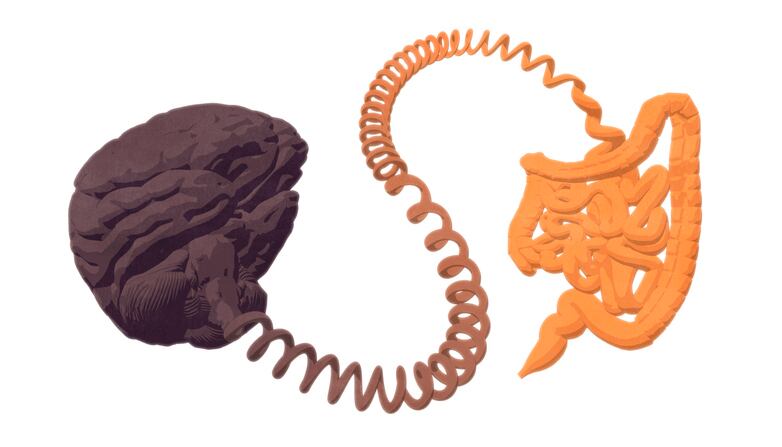
For the last two decades researchers have been building on evidence that focused on key roles that gut microbiota and its metabolites play in controlling glucose metabolism. While researcher’s understanding of gut microbe’s function in controlling glycaemia is still developing, scientists at The Université catholique de Louvain in Belgium have set out to determine whether the modulation of gut microbiota by prebiotics could identify novel local intestinal factors.
In the new report, the identification of one bacteria and/or one of its active metabolites is investigated as a potential novel therapeutic strategy.
Background
In 2004, Claude Knauf (INSERM) and Patrice Cani (UCLouvain) combined forces to understand the mechanisms behind the development of type 2 diabetes, with the ultimate goal being to identify new therapeutic targets. In 2013, they created an international laboratory, 'NeuroMicrobiota Lab' (INSERM-UCLouvain), to identify links between the brain and intestinal bacteria.
Lost in translation
During their research, the team observed that when the gut digests food, it sends a signal to the brain to determine how to handle incoming fats and sugars. The brain then signals various organs (liver, muscles, adipose tissue) to prepare to lower blood sugar and fat levels.
However, this mechanism doesn’t work in a diabetic individuals because the gut malfunctions and doesn’t send the signal to the brain. The miscommunication is a result of the intestine hypercontracting. The commands to get the sugar out of the blood no longer pass, leading to hyperglycemia. The malfunction also means no insulin action is taken, resulting in insulin resistance.
The study
To gain a better understanding of this hypercontractility, the INSERM-UCLouvain researchers analyzed the differences in the intestine’s structure and action of prebiotics within the microbiota in 'normal' and 'diabetic' mice. They noticed that a particular lipid was severely deficient in both diabetic mice and diabetic people. The team therefore tested the impact of the lipid on the use of sugars, on the contraction of the intestine and finally on diabetes.
New Discoveries
Researchers Anne Abot and Eve Wemelle discovered that this specific lipid is the key to restoring the use of sugar by directly acting on the gut. The research has allowed the team to better understand the important role of gut microbiota in altering the production of bioactive lipids, which leads to restoring gut-brain communication.
Key findings
- Modulation of gut microbiota composition by prebiotics improves glucose homeostasis by acting on ENS/duodenal contraction couple.
- The mechanism of prebiotic action implies the presence of an enkephalin/mu-opioid receptor and proliferator-activated receptor γ signaling and a bioactive lipid, 12S-hydroxyeicosatetraenoic acid.
- The expression of enzymes implicated in enteric neurotransmitter synthesis and enkephalin signaling are altered in the duodenum of diabetic human.
Implications
“Using a prebiotic approach, we identified enkephalin and 12-HETE as new enterosynes with potential real beneficial and safety impact in diabetic humans,” the report noted. “This study is the first showing the alteration of ENS and enkephalin signalling in the duodenum of diabetic human. Deciphering the mode of action of gut microbiota on ENS could represent a real safety and innovative therapeutic evidence for patients with diabetic.”
As an added bonus, the INSERM-UCLouvain research team also contributed to the discovery of a new bioactive lipid that reduces intestinal inflammation using the same approach. It is directly produced by certain gut bacteria, also identified in this study and therefore the two approaches, either the lipid or one or more bacteria, could serve as a therapeutic target, according to the report.
The researchers said that treatment possibilities could include modifying the body's production of such lipids or taking them orally, all of which are currently under study.
Source: Gut
Online 05 October 2020. doi: 10.1136/gutjnl-2019-320230
"Identification of new enterosynes using prebiotics: roles of bioactive lipids and mu-opioid receptor signalling in humans and mice"
Authors: A. Abot et al.