Compared to more established categories such as gut health and weight management, respiratory health is a less-explored space, although consumer awareness and interest in this area are on the rise.
Respiratory health concerns in South Korea are strongly influenced by environmental factors, particularly fine dust, which has been recognized as a persistent and significant public health issue in the country.
Seasonal factors also play a role, as respiratory symptoms tend to become more frequent or aggravated during colder periods. Respiratory health topics become more pronounced during periods of elevated fine dust levels.
Additionally, smoking and secondhand smoke are key environmental factors contributing to respiratory burden.
“Fine dust is associated with airway irritation, inflammation, and oxidative stress, and long-term exposure has been linked to reduced lung function and increased respiratory symptoms such as coughing and sputum,” Sol Lee, regional sales manager at CKD Bio, told NutraIngredients.
“In this context, our CKDB-315 may be positioned as a functional ingredient supported by human clinical data that tackles bronchial and respiratory health needs related to environmental exposure.”
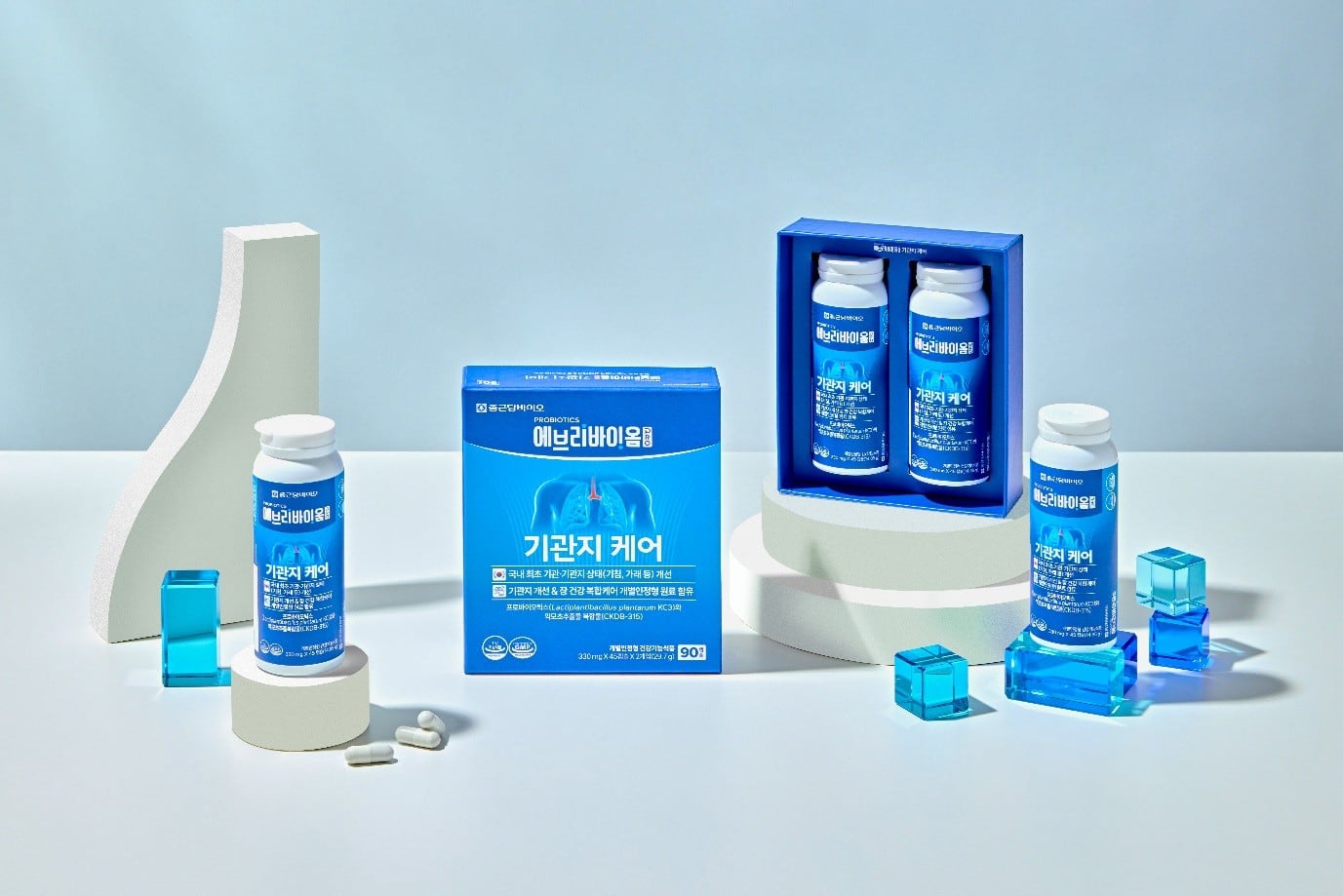
A combination of probiotic and natural extract—considered relatively rare in the Korean market, CKDB-315 was developed based on the gut-lung axis concept and designed to achieve a synergistic effect through complementary mechanisms.
Specifically, the formulation comprises Lactiplantibacillus plantarum KC3 isolated from kimchi and extract from Leonurus japonicus herb (oriental motherwort).
The probiotic strain has shown to exhibit significant immunomodulatory potential, while the Leonurus japonicus extract, sourced from KT&G, is known for its antioxidant and anti-inflammatory properties.
“Such approaches remain limited within the Individually Recognized ingredient segment in the Korean market. To our knowledge, CKDB-315 is the first probiotic ingredient to obtain individual recognition for respiratory health from the Ministry of Food and Drug Safety (MFDS).
“During the clinical study, participants showed strong interest in product availability, frequently asking where it could be purchased. Follow-up inquiries were also received after completion of the study. This suggests that consumer interest in respiratory health solutions is emerging, particularly for products addressing environmental factors such as air pollution.”
In Korea, the ingredient has been applied in a range of finished products launched under CKD Bio’s supplements brand Everibiome.
The company is currently engaged in discussions with domestic pharmaceutical companies and selected international partners for application opportunities, while product introductions and sample evaluations are actively ongoing.
Sustained strength of body fat reduction category
According to Lee, the company’s body fat reduction ingredient, CKDB-322, is undergoing the Individual Recognition review process by the MFDS, with approval expected between mid-May and August 2026.
It is a functional ingredient consisting of a probiotic strain (Lactiplantibacillus plantarum Q180) and a marine microalgae component (Phaeodactylum tricornutum).
While existing body fat reduction ingredients are typically based on single probiotic strain or conventional materials, CKDB-322 represents a structurally differentiated approach.
In a 12-week randomized, double-blind, placebo-controlled clinical trial, CKDB-322 demonstrated statistically significant improvements across multiple adiposity-related indicators, including body fat mass, body fat percentage, BMI, and abdominal fat area, as well as two blood markers.
“The L. plantarum Q180 strain is already recognized in Korea for reducing postprandial blood triglycerides. CKDB-322 further demonstrated reductions in triglycerides and leptin, indicating improvements in both body fat-related parameters and lipid metabolism,” Lee said.
“In addition, the use of marine microalgae provides a plant-based source of omega-3 (EPA), enabling a formulation in which probiotic and microalgae-derived components act together within a single material.”
She added that development at the moment is focused on capsule formulations to ensure stability and controlled delivery.
Body fat reduction is a well-established category within the Korean health functional foods market.
Based on data from the Korea Health Functional Food Association, body fat reduction products accounted for approximately 3.3% of total market value in 2025.
From a supply perspective, body fat reduction is also one of the most laboriously developed categories, with approximately 47 individually recognized ingredients approved between 2004 and 2024, ranking among the top functional categories.
At the same time, consumer perception data show that approximately 26% of respondents are concerned about body fat reduction. Notably, higher levels of interest are observed among consumers in their 20s (33.8%) and 30s (30.9%), as well as among female consumers (29.7%).“These findings indicate that body fat reduction is a category supported by both sustained consumer demand and active ingredient development.”