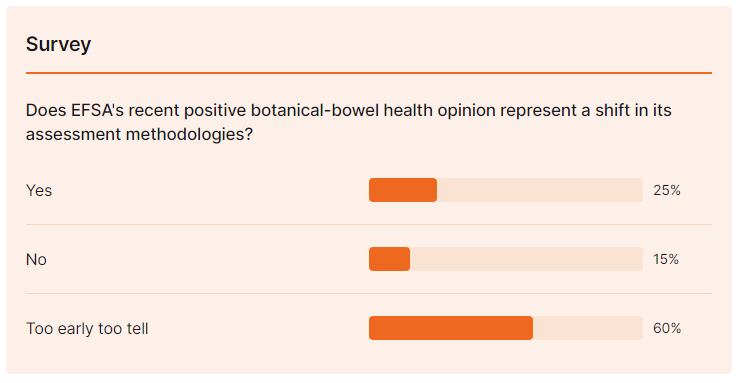
It may be too early to say but there are definitely a couple of encouraging signs for herb players that may be a pointer to how the European Food Safety Authority will eventually deal with some 2078 on-hold health claim submissions when the European Commission and member states can work out what kind of science is good enough to back botanical claims.
Rome-based observer, Luca Bucchini, PhD, from Hylobates Consulting, called the opinion a scientific method sea change.
For most pundits a few things stand out in the article 13.5 opinion issued to French firm Vivatech: 1. Traditional use data is being assessed; 2. Medical data is being assessed; 3. Unpublished data is being assessed.
For the record, EFSA’s Panel on Dietetic Products, Nutrition and Allergies (NDA) said there was sound scientific data to demonstrate short-term constipation alleviation from hydroxyanthracene root and rhizome derivatives in the Vivatech food supplement, Transitech.
The twist being that, drawing on medical trial data, the NDA said the effect was due to hydroxyanthracene alone, not the whole supplement which contained a bunch of other herbal extracts.
Food v medicine
In most EU member states hydroxyanthracene is classified as a medicinal component, not a foodstuff nutrient, although it must be said that there are precedents for this situation in the form of red yeast rice extract, monacolin K, and melatonin – both winners of claims under the nutrition and health claims regulation (NHCR) but not necessarily permitted in foods and food supplements in all EU states due to medical classification.

Dr Robert Verkerk, PhD, executive and scientific director at the Alliance for Natural Health International (ANH-I) had the following to say: “A few things about this positive opinion are at odds with many previous assessments. EFSA has jumped in with hydroxyanthracene derivatives, regarded as medicinal by most EU authorities because of their laxative effect, and seem willing to now see them as food - albeit in limited concentrations.”
“The data that clinched the opinion was basically one unpublished trial, also unusual. But then the parameters were limited, straight forward and clearly defined. But of course EFSA's opinion means nothing unless it gets through the Commission and is successfully added to the authorised list of health claims.”
There are no guarantees there.
Old and new traditions
European Botanical Forum’s Patrick Coppens ventured: “It is not sufficient yet to firmly say that EFSA appears to be willing to consider tradition of use, but it clearly indicates that EFSA acknowledges that generally accepted scientific evidence includes opinions from the European Medicines Agency Committee on Herbal Medicinal Products (HMPC) relating to well-established use – which of course still ranks a bit higher on the scale of evidence than tradition of use.”
“Nevertheless it is very positive that the EFSA experts are considering as valid opinions occasions where botanical experts have agreed on the plausibility of an effect. We hope that they will also do so when the experts have agreed on such plausibility on the basis of observational evidence and experience of use.”
Watch this space to so see of this offers real hope in the months and years ahead for botanicals players in Europe and abroad…
(As an aside, strangely, Vivatech appears to have vanished from the face of the Earth despite the rare bounty of an EFSA-backed botanical health claim.)