Italy cracks whip on health claim abusers - fines could reach €5m

One pundit praised the regulator while wondering if the whip cracking is enough to sway a market that has long-exercised marketing alacrity over solid scientific data, or as the pundit put it, a tendency toward “guru” science.
“There are two questions,” stated EU food law expert Luca Bucchini. “First will firms be able to build a business model around authorised health claims, replacing the current curative/guru-scientist food supplement model? I would advise such companies to think hard about alternatives. Second, will authorities in Italy and elsewhere keep up the pressure?"
The fines could in theory go as high as €5m under Italian law.
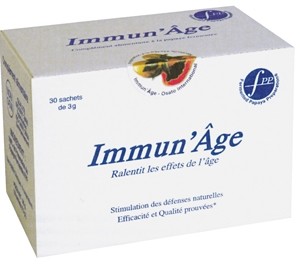
Recent censorings include Named Spa, a significant €33m Italian player, which Italy’s Market and Competition Authority fined €250,000 for exaggerated and unauthorised immunity claims for a fermented papaya extract called Immun’Âge. The claims dated back to 2007.
That fine is believed to be the biggest ever administered for a breach of the EU nutrition and health claims regulation (NHCR) in any of the European Union’s 28 member states.
The Italian authority previously fined Humana, another local firm, €110,000 for similar breaches for some of its food supplements.
Neither company has spoken of the fines, or its post-fine intentions, but Rome-based consultant Bucchini said the firms were asking for trouble with marketing that was clearly outside EU law – especially after the imposition of the NHCR in 2008 and the bulk publication of authorised/non-authorised article 13.1 claims at the back-end of 2012.
License to claim
"Named is well-known for its marketing style, and, though the fine is a record and shows Italy can be tough on food supplements, it is remarkable that it took seven years for the authorities to act,” Bucchini said.
“In general, surprising as it seems there are still companies relying on isolated studies or opinions of scientists for their communication. Some marketeers, managers and scientists continue not to accept that, like driving, making health claims requires a license: It does not matter if you are good driver, but it matters whether you have a license.”
“In the case of Named, of course, the messages were not in line with mainstream science, and many claims were medicinal. They are well known for their aggressive communication.”
The UK Advertising Standards Authority (ASA) has also drawn attention for the strictness of its rulings, but it is yet to issue fines against claims offenders, preferring to name and shame them into compliance.